Abnormal development of the olfactory bulb and reproductive system in mice lacking prokineticin receptor PKR2
- PMID: 16537498
- PMCID: PMC1449660
- DOI: 10.1073/pnas.0508881103
Abnormal development of the olfactory bulb and reproductive system in mice lacking prokineticin receptor PKR2
Abstract
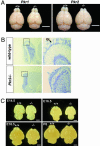
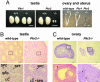
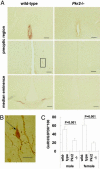
Prokineticins, multifunctional secreted proteins, activate two endogenous G protein-coupled receptors PKR1 and PKR2. From in situ analysis of the mouse brain, we discovered that PKR2 is predominantly expressed in the olfactory bulb (OB). To examine the role of PKR2 in the OB, we created PKR1- and PKR2-gene-disrupted mice (Pkr1(-/-) and Pkr2(-/-), respectively). Phenotypic analysis indicated that not Pkr1(-/-)but Pkr2(-/-)mice exhibited hypoplasia of the OB. This abnormality was observed in the early developmental stages of fetal OB in the Pkr2(-/-) mice. In addition, the Pkr2(-/-) mice showed severe atrophy of the reproductive system, including the testis, ovary, uterus, vagina, and mammary gland. In the Pkr2(-/-) mice, the plasma levels of testosterone and follicle-stimulating hormone were decreased, and the mRNA transcription levels of gonadotropin-releasing hormone in the hypothalamus and luteinizing hormone and follicle-stimulating hormone in the pituitary were also significantly reduced. Immunohistochemical analysis revealed that gonadotropin-releasing hormone neurons were absent in the hypothalamus in the Pkr2(-/-) mice. The phenotype of the Pkr2(-/-) mice showed similarity to the clinical features of Kallmann syndrome, a human disease characterized by association of hypogonadotropic hypogonadism and anosmia. Our current findings demonstrated that physiological activation of PKR2 is essential for normal development of the OB and sexual maturation.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures

References
-
- Li M., Bullock C. M., Knauer D. J., Ehlert F. J., Zhou Q. Y. Mol. Pharmacol. 2001;59:692–698. - PubMed
-
- Bullock C. M., Li J. D., Zhou Q. Y. Mol. Pharmacol. 2004;65:582–588. - PubMed
-
- Lin D. C., Bullock C. M., Ehlert F. J., Chen J. L., Tian H., Zhou Q. Y. J. Biol. Chem. 2002;277:19276–19280. - PubMed
-
- Masuda Y., Takatsu Y., Terao Y., Kumano S., Ishibashi Y., Suenaga M., Abe M., Fukusumi S., Watanabe T., Shintani Y., et al. Biochem. Biophys. Res. Commun. 2002;293:396–402. - PubMed
-
- Soga T., Matsumoto S., Oda T., Saito T., Hiyama H., Takasaki J., Kamohara M., Ohishi T., Matsushime H., Furuichi K. Biochim. Biophys. Acta. 2002;1579:173–179. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

