Identification of proangiogenic genes and pathways by high-throughput functional genomics: TBK1 and the IRF3 pathway
- PMID: 16537515
- PMCID: PMC1449677
- DOI: 10.1073/pnas.0511319103
Identification of proangiogenic genes and pathways by high-throughput functional genomics: TBK1 and the IRF3 pathway
Abstract
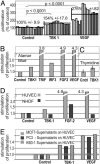
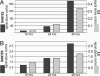
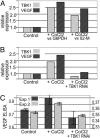
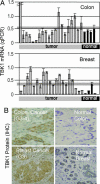
A genome-wide phenotype screen was used to identify factors and pathways that induce proliferation of human umbilical vein endothelial cells (HUVEC). HUVEC proliferation is a recognized marker for factors that modulate vascularization. Screening "hits" included known proangiogenic factors, such as VEGF, FGF1, and FGF2 and additional factors for which a direct association with angiogenesis was not previously described. These include the kinase TBK1 as well as Toll-like receptor adaptor molecule and IFN regulatory factor 3. All three proteins belong to one signaling pathway that mediates induction of gene expression, including a mixture of secreted factors, which, in concert, mediate proliferative activity toward endothelial cells. TBK1 as the "trigger" of this pathway is induced under hypoxic conditions and expressed at significant levels in many solid tumors. This pattern of expression and the decreased expression of angiogenic factors in cultured cells upon RNA-interference-mediated ablation suggests that TBK1 is important for vascularization and subsequent tumor growth and a target for cancer therapy.
Conflict of interest statement
Conflict of interest statement: C.K., H.G., R.S., K.K.-H., and U.B. were or are employees of Xantos Biomedicine.
Figures

Similar articles
-
Suppression of MyD88- and TRIF-dependent signaling pathways of Toll-like receptor by (-)-epigallocatechin-3-gallate, a polyphenol component of green tea.Biochem Pharmacol. 2006 Sep 28;72(7):850-9. doi: 10.1016/j.bcp.2006.06.021. Epub 2006 Aug 4. Biochem Pharmacol. 2006. PMID: 16890209
-
Evaluating TBK1 as a therapeutic target in cancers with activated IRF3.Mol Cancer Res. 2014 Jul;12(7):1055-66. doi: 10.1158/1541-7786.MCR-13-0642. Epub 2014 Apr 21. Mol Cancer Res. 2014. PMID: 24752990
-
TBK1-targeted suppression of TRIF-dependent signaling pathway of toll-like receptor 3 by auranofin.Arch Pharm Res. 2010 Jun;33(6):939-45. doi: 10.1007/s12272-010-0618-2. Epub 2010 Jul 6. Arch Pharm Res. 2010. PMID: 20607500
-
Both protein activation and gene expression are involved in early vascular tube formation in vitro.Clin Cancer Res. 2002 Sep;8(9):3019-26. Clin Cancer Res. 2002. PMID: 12231549
-
Regulation and function of IKK and IKK-related kinases.Sci STKE. 2006 Oct 17;2006(357):re13. doi: 10.1126/stke.3572006re13. Sci STKE. 2006. PMID: 17047224 Review.
Cited by
-
RAMP1 is a direct NKX3.1 target gene up-regulated in prostate cancer that promotes tumorigenesis.Am J Pathol. 2013 Sep;183(3):951-63. doi: 10.1016/j.ajpath.2013.05.021. Epub 2013 Jul 16. Am J Pathol. 2013. PMID: 23867798 Free PMC article.
-
NF-κB and cancer: a paradigm of Yin-Yang.Am J Cancer Res. 2011;1(2):192-221. Epub 2010 Dec 6. Am J Cancer Res. 2011. PMID: 21969033 Free PMC article.
-
Negative regulation of TBK1-mediated antiviral immunity.FEBS Lett. 2013 Mar 18;587(6):542-8. doi: 10.1016/j.febslet.2013.01.052. Epub 2013 Feb 8. FEBS Lett. 2013. PMID: 23395611 Free PMC article. Review.
-
Axl-mediated activation of TBK1 drives epithelial plasticity in pancreatic cancer.JCI Insight. 2019 Apr 2;5(9):e126117. doi: 10.1172/jci.insight.126117. JCI Insight. 2019. PMID: 30938713 Free PMC article.
-
Elevated expression of TANK-binding kinase 1 enhances tamoxifen resistance in breast cancer.Proc Natl Acad Sci U S A. 2014 Feb 4;111(5):E601-10. doi: 10.1073/pnas.1316255111. Epub 2014 Jan 21. Proc Natl Acad Sci U S A. 2014. PMID: 24449872 Free PMC article.
References
-
- Lander E. S., Linton L. M., Birren B., Nusbaum C., Zoby M. C., Baldwin J., Devon K., Dewar K., Doyle M., FitzHugh W., et al. Nature. 2001;409:860–921. errata (2001) 411, 720 and (2001) 412, 565. - PubMed
-
- Venter J. C., Adams M. D., Myers E. W., Li P. W., Mural R. J., Sutton G. G., Smith H. O., Yandell M., Evans C. A., Holt R. A., et al. Science. 2001;291:1304–1351. erratum (2001) 292, 1838. - PubMed
-
- Boguski M. S., Lowe T. M., Tolstoshev C. M. Nat. Genet. 1993;4:332–333. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

