Membrane-anchored inhibitory peptides capture human immunodeficiency virus type 1 gp41 conformations that engage the target membrane prior to fusion
- PMID: 16537592
- PMCID: PMC1440409
- DOI: 10.1128/JVI.80.7.3249-3258.2006
Membrane-anchored inhibitory peptides capture human immunodeficiency virus type 1 gp41 conformations that engage the target membrane prior to fusion
Abstract
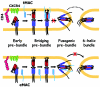
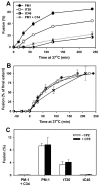
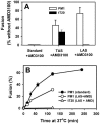
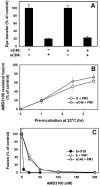
Soluble peptides derived from the C-terminal heptad repeat domain of human immunodeficiency virus type 1 (HIV-1) gp41 are potent inhibitors of HIV-1 entry and gp41-induced fusion. Target membrane-anchored variants of these peptides have been shown to retain inhibitory activity. Both soluble and membrane-anchored C peptides (MACs) are thought to block fusion by binding to the N-terminal coiled coil domain of gp41 and preventing formation of the final six-helix bundle structure. However, interactions of target MACs with gp41 must be restricted to a subset of trimers that have their hydrophobic fusion peptides inserted into the target membrane. This unique feature of MACs was used to identify the intermediate step of fusion at which gp41 engaged the target membrane. Fusion between HIV envelope-expressing effector cells and target cells was measured by fluorescence microscopy. Expression of MACs in target cells led to less than twofold reduction in the extent of fusion. However, when reaction was first arrested by adding lysolipids that disfavored membrane merger, and the lipids were subsequently removed by washing, control cells supported fusion, whereas those that expressed MACs did not. The drastically improved potency of MACs implies that, at lipid-arrested stage, gp41 bridges the viral and target cell membranes and therefore more optimally binds the membrane-anchored peptides. Experimental demonstration of this intermediate shows that, similar to fusion induced by many other viral glycoproteins, engaging the target membrane by HIV-1 gp41 permits coupling between six-helix bundle formation and membrane merger.
Figures


References
-
- Binley, J. M., R. W. Sanders, B. Clas, N. Schuelke, A. Master, Y. Guo, F. Kajumo, D. J. Anselma, P. J. Maddon, W. C. Olson, and J. P. Moore. 2000. A recombinant human immunodeficiency virus type 1 envelope glycoprotein complex stabilized by an intermolecular disulfide bond between the gp120 and gp41 subunits is an antigenic mimic of the trimeric virion-associated structure. J. Virol. 74:627-643. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

