Effects of type I interferons on Friend retrovirus infection
- PMID: 16537611
- PMCID: PMC1440373
- DOI: 10.1128/JVI.80.7.3438-3444.2006
Effects of type I interferons on Friend retrovirus infection
Erratum in
- J Virol. 2007 Jun;81(11):6160
Abstract
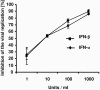
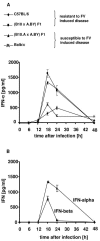
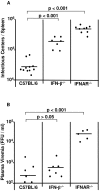
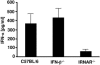
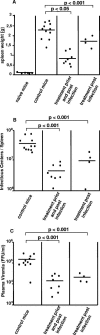
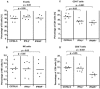
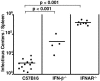
The type I interferon (IFN) response plays an important role in the control of many viral infections. However, since there is no rodent animal model for human immunodeficiency virus, the antiviral effect of IFN-alpha and IFN-beta in retroviral infections is not well characterized. In the current study we have used the Friend virus (FV) model to determine the activity of type I interferons against a murine retrovirus. After FV infection of mice, IFN-alpha and IFN-beta could be measured between 12 and 48 h in the serum. The important role of type I IFN in the early immune defense against FV became evident when mice deficient in IFN type I receptor (IFNAR(-/-)) or IFN-beta (IFN-beta(-/-)) were infected. The levels of FV infection in plasma and in spleen were higher in both strains of knockout mice than in C57BL/6 wild-type mice. This difference was induced by an antiviral effect of IFN-alpha and IFN-beta and was most likely mediated by antiviral enzymes as well as by an effect of these IFNs on T-cell responses. Interestingly, the lack of IFNAR and IFN-beta enhanced viral loads during acute and chronic FV infection. Exogenous IFN-alpha could be used therapeutically to reduce FV replication during acute but not chronic infection. These findings indicate that type I IFN plays an important role in the immediate antiviral defense against Friend retrovirus infection.
Figures
Similar articles
-
Role of interleukin-4 (IL-4), IL-12, and gamma interferon in primary and vaccine-primed immune responses to Friend retrovirus infection.J Virol. 2001 Jan;75(2):654-60. doi: 10.1128/JVI.75.2.654-660.2001. J Virol. 2001. PMID: 11134279 Free PMC article.
-
Immunotherapy With Interferon α11, But Not Interferon Beta, Controls Persistent Retroviral Infection.Front Immunol. 2022 Jan 20;12:809774. doi: 10.3389/fimmu.2021.809774. eCollection 2021. Front Immunol. 2022. PMID: 35126368 Free PMC article.
-
Temporal effects of gamma interferon deficiency on the course of Friend retrovirus infection in mice.J Virol. 2002 Mar;76(5):2225-32. doi: 10.1128/jvi.76.5.2225-2232.2002. J Virol. 2002. PMID: 11836400 Free PMC article.
-
Host genetic factors that control immune responses to retrovirus infections.Vaccine. 2008 Jun 6;26(24):2981-96. doi: 10.1016/j.vaccine.2008.01.004. Epub 2008 Jan 22. Vaccine. 2008. PMID: 18255203 Review.
-
Immune control and prevention of chronic Friend retrovirus infection.Front Biosci. 2007 Jan 1;12:1544-51. doi: 10.2741/2167. Front Biosci. 2007. PMID: 17127401 Review.
Cited by
-
Lentiviral Protein Transfer Vectors Are an Efficient Vaccine Platform and Induce a Strong Antigen-Specific Cytotoxic T Cell Response.J Virol. 2015 Sep;89(17):9044-60. doi: 10.1128/JVI.00844-15. Epub 2015 Jun 17. J Virol. 2015. PMID: 26085166 Free PMC article.
-
Use of recombinant interferon omega in feline retrovirosis: from theory to practice.Vet Immunol Immunopathol. 2011 Oct 15;143(3-4):301-6. doi: 10.1016/j.vetimm.2011.06.008. Epub 2011 Jun 12. Vet Immunol Immunopathol. 2011. PMID: 21719116 Free PMC article.
-
HIV-1 Infection and Type 1 Interferon: Navigating Through Uncertain Waters.AIDS Res Hum Retroviruses. 2019 Jan;35(1):25-32. doi: 10.1089/AID.2018.0161. Epub 2018 Aug 15. AIDS Res Hum Retroviruses. 2019. PMID: 29999412 Free PMC article. Review.
-
NK cells improve control of friend virus infection in mice persistently infected with murine cytomegalovirus.Retrovirology. 2013 Jun 5;10:58. doi: 10.1186/1742-4690-10-58. Retrovirology. 2013. PMID: 23738889 Free PMC article.
-
Type I Interferons Act Directly on Nociceptors to Produce Pain Sensitization: Implications for Viral Infection-Induced Pain.J Neurosci. 2020 Apr 29;40(18):3517-3532. doi: 10.1523/JNEUROSCI.3055-19.2020. Epub 2020 Apr 3. J Neurosci. 2020. PMID: 32245829 Free PMC article.
References
-
- Ankel, H., R. M. Capobianchi, C. Castiletti, and F. Dianzanni. 1994. Interferon induction by HIV glycoprotein 120: role of the V3 loop. J. Interferon Res. 14:S209-P8-4. - PubMed
-
- Biron, C. A. 1998. Role of early cytokines, including alpha and beta interferons (IFN-α/β), in innate and adaptive immune responses to viral infections. Immunology 10:383-390. - PubMed
-
- Deonarain, R., D. Cerullo, K. Fuse, P. P. Liu, and E. N. Fish. 2004. Protective role for interferon-beta in coxsackievirus B3 infection. Circulation 110:3540-3543. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

