Infection and persistence of rhesus monkey rhadinovirus in immortalized B-cell lines
- PMID: 16537632
- PMCID: PMC1440374
- DOI: 10.1128/JVI.80.7.3644-3649.2006
Infection and persistence of rhesus monkey rhadinovirus in immortalized B-cell lines
Abstract
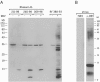
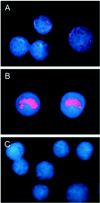
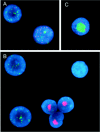
Similar to its close relative human herpesvirus 8, rhesus monkey rhadinovirus (RRV) persists predominantly in B cells of its natural host. Rhesus monkey B-cell lines immortalized by the Epstein-Barr-related virus from rhesus monkeys (rhEBV) were used as targets for infection by RRV. These cultured B cells were susceptible to infection by RRV and continued to produce low titers of RRV for months of continuous culture. Infection by RRV did not detectably alter the growth rates of these B-cell lines when it was measured at standard or reduced serum concentrations. Depending on the cell line, 5 to 40% of the B cells stained positive for the RRV genome by fluorescence in situ hybridization (FISH). Most RRV-positive cells showed a fine punctate nuclear staining pattern consistent with latent infection, while a small minority of cells (0.2 to 1%) contained large, intensely staining nuclear foci consistent with productive, replicative infection. Greater than 90% of the cells were rhEBV genome positive in a pattern consistent with latent infection, and again only a small minority of cells showed a productive, replicative staining pattern. Dual, two-color FISH staining revealed coinfection of numerous cells with both RRV and rhEBV, but productive replication of RRV and rhEBV was always observed in separate cells, never in the same cell. Thus, productive replication of RRV is unlinked to that of rhEBV; factors that influence activation to productive replication act separately on RRV and rhEBV, even within the same cell. The percentage of B cells expressing green fluorescent protein (GFP) early after infection with a recombinant RRV containing a GFP reporter gene was dose dependent and at a low multiplicity of infection increased progressively over time until 14 to 17 days after infection. These results establish a naturalistic cell culture system for the study of infection and persistence by RRV in rhesus monkey B cells.
Figures

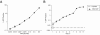
Similar articles
-
A genetic system for rhesus monkey rhadinovirus: use of recombinant virus to quantitate antibody-mediated neutralization.J Virol. 2006 Feb;80(3):1549-62. doi: 10.1128/JVI.80.3.1549-1562.2006. J Virol. 2006. PMID: 16415030 Free PMC article.
-
Rhesus rhadinovirus establishes a latent infection in B lymphocytes in vivo.J Virol. 1999 Sep;73(9):7874-6. doi: 10.1128/JVI.73.9.7874-7876.1999. J Virol. 1999. PMID: 10438883 Free PMC article.
-
Rhesus monkey rhadinovirus (RRV): construction of a RRV-GFP recombinant virus and development of assays to assess viral replication.Virology. 2003 Jul 20;312(1):122-34. doi: 10.1016/s0042-6822(03)00195-8. Virology. 2003. PMID: 12890626
-
Rhesus monkey rhadinovirus: a model for the study of KSHV.Curr Top Microbiol Immunol. 2007;312:43-69. doi: 10.1007/978-3-540-34344-8_2. Curr Top Microbiol Immunol. 2007. PMID: 17089793 Review.
-
Rhesus macaque rhadinovirus-associated disease.Curr Opin Virol. 2013 Jun;3(3):245-50. doi: 10.1016/j.coviro.2013.05.016. Epub 2013 Jun 6. Curr Opin Virol. 2013. PMID: 23747119 Free PMC article. Review.
Cited by
-
Macaque homologs of Kaposi's sarcoma-associated herpesvirus (KSHV) infect germinal center lymphoid cells, epithelial cells in skin and gastrointestinal tract and gonadal germ cells in naturally infected macaques.Virology. 2018 Jun;519:106-120. doi: 10.1016/j.virol.2018.04.007. Epub 2018 Apr 22. Virology. 2018. PMID: 29689462 Free PMC article.
-
Viral FLICE inhibitory protein of rhesus monkey rhadinovirus inhibits apoptosis by enhancing autophagosome formation.PLoS One. 2012;7(6):e39438. doi: 10.1371/journal.pone.0039438. Epub 2012 Jun 20. PLoS One. 2012. PMID: 22745754 Free PMC article.
-
Progressive accumulation of activated ERK2 within highly stable ORF45-containing nuclear complexes promotes lytic gammaherpesvirus infection.PLoS Pathog. 2014 Apr 10;10(4):e1004066. doi: 10.1371/journal.ppat.1004066. eCollection 2014 Apr. PLoS Pathog. 2014. PMID: 24722398 Free PMC article.
-
Use of a gamma-2 herpesvirus as a vector to deliver antibodies to rhesus monkeys.Gene Ther. 2017 Aug;24(8):487-492. doi: 10.1038/gt.2017.49. Epub 2017 Jun 29. Gene Ther. 2017. PMID: 28660888 Free PMC article.
-
Two herpesviral noncoding PAN RNAs are functionally homologous but do not associate with common chromatin loci.PLoS Pathog. 2018 Nov 1;14(11):e1007389. doi: 10.1371/journal.ppat.1007389. eCollection 2018 Nov. PLoS Pathog. 2018. PMID: 30383841 Free PMC article.
References
-
- Alexander, L., L. Denekamp, A. Knapp, M. R. Auerbach, B. Damania, and R. C. Desrosiers. 2000. The primary sequence of rhesus monkey rhadinovirus isolate 26-95: sequence similarities to Kaposi's sarcoma-associated herpesvirus and rhesus monkey rhadinovirus isolate 17577. J. Virol. 74:3388-3398. - PMC - PubMed
-
- Cesarman, E., Y. Chang, P. S. Moore, J. W. Said, and D. M. Knowles. 1995. Kaposi's sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N. Engl. J. Med. 332:1186-1191. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

