TNF downmodulates the function of human CD4+CD25hi T-regulatory cells
- PMID: 16537805
- PMCID: PMC1895836
- DOI: 10.1182/blood-2005-11-4567
TNF downmodulates the function of human CD4+CD25hi T-regulatory cells
Abstract
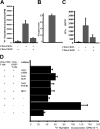
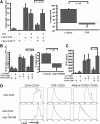
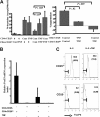
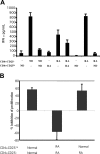
CD4+CD25+ T-regulatory cells (Tregs) play an essential role in maintaining immunologic homeostasis and preventing autoimmunity. However, little is known about the exogenous factors that regulate their differentiation and function. Here, we report that TNF inhibits the suppressive function of both naturally occurring CD4+CD25+ Tregs and TGFbeta1-induced CD4+CD25+ T-regulatory cells. The mechanism of this inhibition involves signaling through TNFRII that is constitutively expressed selectively on unstimulated Tregs and that is up-regulated by TNF. TNF-mediated inhibition of suppressive function is related to a decrease in FoxP3 mRNA and protein expression by the Tregs. Notably, CD4+CD25hi Tregs isolated from patients with active rheumatoid arthritis (RA) expressed reduced levels of FoxP3 mRNA and protein and poorly suppressed the proliferation and cytokine secretion of CD4+ effector T cells in vitro. Treatment with anti-TNF antibody (infliximab) increased FOXP3 mRNA and protein expression by CD4+CD25hi Tregs and restored their suppressive function. Thus, TNF has a novel action in modulating autoimmunity, by inhibiting CD4+CD25+ Treg activity.
Figures




References
-
- Kappler JW, Roehm N, Marrack P. T cell tolerance by clonal elimination in the thymus. Cell. 1987;49: 273-280. - PubMed
-
- Van Parijs L, Abbas AK. Homeostasis and selftolerance in the immune system: turning lymphocytes off. Science. 1998;280: 243-248. - PubMed
-
- Sakaguchi S. Regulatory T cells: key controllers of immunologic self-tolerance. Cell. 2000;101: 455-458. - PubMed
-
- Shevach EM. CD4+ CD25+ suppressor T cells: more questions than answers. Nat Rev Immunol. 2002;2: 389-400. - PubMed
-
- Sakaguchi S. Naturally arising CD4+ regulatory T cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol. 2004;22: 531-562. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

