Cyclin D3 maintains growth-inhibitory activity of C/EBPalpha by stabilizing C/EBPalpha-cdk2 and C/EBPalpha-Brm complexes
- PMID: 16537903
- PMCID: PMC1430318
- DOI: 10.1128/MCB.26.7.2570-2582.2006
Cyclin D3 maintains growth-inhibitory activity of C/EBPalpha by stabilizing C/EBPalpha-cdk2 and C/EBPalpha-Brm complexes
Abstract
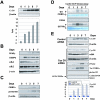
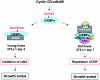
C/EBPalpha arrests proliferation of young livers by inhibition of cdk2. In old mice, C/EBPalpha inhibits growth by repression of E2F-dependent promoters through the C/EBPalpha-Brm complex. In this paper, we show that cyclin D3-cdk4/cdk6 supports the ability of C/EBPalpha to inhibit liver proliferation in both age groups. Although cyclin D3-cdk4/cdk6 kinases are involved in the promotion of growth, they are expressed in terminally differentiated cells, suggesting that they have additional functions in these settings. We demonstrate that C/EBPalpha represents a target for phosphorylation by cyclin D3-cdk4/cdk6 complexes in differentiated liver cells and in differentiated adipocytes. Cyclin D3-cdk4/cdk6 specifically phosphorylate C/EBPalpha at Ser193 in vitro and in the liver and support growth-inhibitory C/EBPalpha-cdk2 and C/EBPalpha-Brm complexes. We found that cyclin D3 is increased in old livers and activates cdk4/cdk6, resulting in stabilization of the C/EBPalpha-Brm complex. Old livers fail to reduce the activity of cyclin D3-cdk4/cdk6 after partial hepatectomy, leading to high levels of C/EBPalpha-Brm complexes after partial hepatectomy, which correlate with weak proliferation. We examined the role of cyclin D3 in the stabilization of C/EBPalpha-cdk2 and C/EBPalpha-Brm by using 3T3-L1 differentiated cells. In these cells, cyclin D3 is increased during differentiation and phosphorylates C/EBPalpha at Ser193, leading to the formation of growth-inhibitory C/EBPalpha-cdk2 and C/EBPalpha-Brm complexes. The inhibition of cyclin D3 blocks the formation of these complexes. Thus, these studies provide a new function of cyclin D3, which is to support the growth-inhibitory activity of C/EBPalpha.
Figures





Similar articles
-
Growth hormone corrects proliferation and transcription of phosphoenolpyruvate carboxykinase in livers of old mice via elimination of CCAAT/enhancer-binding protein alpha-Brm complex.J Biol Chem. 2007 Jan 12;282(2):1468-78. doi: 10.1074/jbc.M608226200. Epub 2006 Nov 15. J Biol Chem. 2007. PMID: 17107955
-
Ghrelin receptor (GHS-R1A) agonists show potential as interventive agents during aging.Ann N Y Acad Sci. 2007 Nov;1119:147-64. doi: 10.1196/annals.1404.023. Ann N Y Acad Sci. 2007. PMID: 18056963 Review.
-
HDAC1 cooperates with C/EBPalpha in the inhibition of liver proliferation in old mice.J Biol Chem. 2008 Sep 19;283(38):26169-78. doi: 10.1074/jbc.M803544200. Epub 2008 Jul 11. J Biol Chem. 2008. PMID: 18622015 Free PMC article.
-
C/EBPalpha arrests cell proliferation through direct inhibition of Cdk2 and Cdk4.Mol Cell. 2001 Oct;8(4):817-28. doi: 10.1016/s1097-2765(01)00366-5. Mol Cell. 2001. PMID: 11684017
-
GSK3beta-cyclin D3-CUGBP1-eIF2 pathway in aging and in myotonic dystrophy.Cell Cycle. 2009 Aug;8(15):2356-9. doi: 10.4161/cc.8.15.9248. Epub 2009 Aug 11. Cell Cycle. 2009. PMID: 19571675 Review.
Cited by
-
The miRNA Contribution in Adipocyte Maturation.Noncoding RNA. 2024 Jun 12;10(3):35. doi: 10.3390/ncrna10030035. Noncoding RNA. 2024. PMID: 38921832 Free PMC article.
-
Elimination of Age-Associated Hepatic Steatosis and Correction of Aging Phenotype by Inhibition of cdk4-C/EBPα-p300 Axis.Cell Rep. 2018 Aug 7;24(6):1597-1609. doi: 10.1016/j.celrep.2018.07.014. Cell Rep. 2018. PMID: 30089269 Free PMC article.
-
Catabolism of Branched Chain Amino Acids Contributes Significantly to Synthesis of Odd-Chain and Even-Chain Fatty Acids in 3T3-L1 Adipocytes.PLoS One. 2015 Dec 28;10(12):e0145850. doi: 10.1371/journal.pone.0145850. eCollection 2015. PLoS One. 2015. PMID: 26710334 Free PMC article.
-
pRb-dependent cyclin D3 protein stabilization is required for myogenic differentiation.Mol Cell Biol. 2007 Oct;27(20):7248-65. doi: 10.1128/MCB.02199-06. Epub 2007 Aug 20. Mol Cell Biol. 2007. PMID: 17709384 Free PMC article.
-
Regulation of apoptotic and growth inhibitory activities of C/EBPalpha in different cell lines.Exp Cell Res. 2008 Apr 15;314(7):1626-39. doi: 10.1016/j.yexcr.2008.01.028. Epub 2008 Feb 13. Exp Cell Res. 2008. PMID: 18313667 Free PMC article.
References
-
- Bartkova, J., J. Lukas, M. Strauss, and J. Bartek. 1998. Cyclin D3: requirement for G1/S transition and high abundance in quiescent tissues suggest a dual role in proliferation and differentiation. Oncogene 17:1027-1037. - PubMed
-
- Conboy, I. M., M. J. Conboy, A. J. Wagers, E. R. Girma, I. L. Weisman, and T. A. Rando. 2005. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 43:760-764. - PubMed
-
- de La Serna, I. L., K. Roy, K. A. Carlson, and A. N. Imbalzano. 2001. MyoD can induce Cell cycle arrest but not muscle differentiation in the presence of dominant negative SWI/SNF chromatin remodeling enzymes. J. Biol. Chem. 276:41486-41491. - PubMed
-
- Doglioni, C., C. Chiarelli, E. Macri, A. P. Dei Tos, E. Meggiolaro, P. Dalla Palma, and M. Barbaresch. 1998. Cyclin D3 expression in normal, reactive and neoplastic tissues. J. Pathol. 185:159-166. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
