Modulation of the transcription regulatory program in yeast cells committed to sporulation
- PMID: 16542486
- PMCID: PMC1557749
- DOI: 10.1186/gb-2006-7-3-r20
Modulation of the transcription regulatory program in yeast cells committed to sporulation
Abstract
Background: Meiosis in budding yeast is coupled to the process of sporulation, where the four haploid nuclei are packaged into a gamete. This differentiation process is characterized by a point of transition, termed commitment, when it becomes independent of the environment. Not much is known about the mechanisms underlying commitment, but it is often assumed that positive feedback loops stabilize the underlying gene-expression cascade.
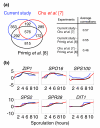
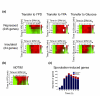
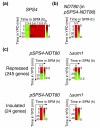
Results: We describe the gene-expression program of committed cells. Sporulating cells were transferred back to growth medium at different stages of the process, and their transcription response was characterized. Most sporulation-induced genes were immediately downregulated upon transfer, even in committed cells that continued to sporulate. Focusing on the metabolic-related transcription response, we observed that pre-committed cells, as well as mature spores, responded to the transfer to growth medium in essentially the same way that vegetative cells responded to glucose. In contrast, committed cells elicited a dramatically different response.
Conclusion: Our results suggest that cells ensure commitment to sporulation not by stabilizing the process, but by modulating their gene-expression program in an active manner. This unique transcriptional program may optimize sporulation in an environment-specific manner.
Figures



Similar articles
-
Saccharomyces cerevisiae exhibits a sporulation-specific temporal pattern of transcript accumulation.Mol Cell Biol. 1985 Apr;5(4):751-61. doi: 10.1128/mcb.5.4.751-761.1985. Mol Cell Biol. 1985. PMID: 3887135 Free PMC article.
-
Regulation of sporulation in the yeast Saccharomyces cerevisiae.Acta Biochim Pol. 2010;57(3):241-50. Acta Biochim Pol. 2010. PMID: 20842291 Review.
-
RSC Nucleosome-remodeling complex plays prominent roles in transcriptional regulation throughout budding yeast gametogenesis.Biosci Biotechnol Biochem. 2004 Apr;68(4):909-19. doi: 10.1271/bbb.68.909. Biosci Biotechnol Biochem. 2004. PMID: 15118322
-
Isolation of genes expressed preferentially during sporulation in the yeast Saccharomyces cerevisiae.Proc Natl Acad Sci U S A. 1983 May;80(10):3000-4. doi: 10.1073/pnas.80.10.3000. Proc Natl Acad Sci U S A. 1983. PMID: 6304689 Free PMC article.
-
Genome reprogramming during sporulation.Int J Dev Biol. 2009;53(2-3):425-32. doi: 10.1387/ijdb.082687jg. Int J Dev Biol. 2009. PMID: 19412896 Free PMC article. Review.
Cited by
-
Unresolved Recombination Intermediates Cause a RAD9-Dependent Cell Cycle Arrest in Saccharomyces cerevisiae.Genetics. 2019 Nov;213(3):805-818. doi: 10.1534/genetics.119.302632. Epub 2019 Sep 27. Genetics. 2019. PMID: 31562181 Free PMC article.
-
Spore germination in Saccharomyces cerevisiae: global gene expression patterns and cell cycle landmarks.Genome Biol. 2007;8(11):R241. doi: 10.1186/gb-2007-8-11-r241. Genome Biol. 2007. PMID: 17999778 Free PMC article.
-
The Ras/cAMP pathway and the CDK-like kinase Ime2 regulate the MAPK Smk1 and spore morphogenesis in Saccharomyces cerevisiae.Genetics. 2009 Feb;181(2):511-23. doi: 10.1534/genetics.108.098434. Epub 2008 Dec 15. Genetics. 2009. PMID: 19087957 Free PMC article.
-
Nutrient Signaling, Stress Response, and Inter-organelle Communication Are Non-canonical Determinants of Cell Fate.Cell Rep. 2020 Dec 1;33(9):108446. doi: 10.1016/j.celrep.2020.108446. Cell Rep. 2020. PMID: 33264609 Free PMC article.
-
Elevated mutation rate during meiosis in Saccharomyces cerevisiae.PLoS Genet. 2015 Jan 8;11(1):e1004910. doi: 10.1371/journal.pgen.1004910. eCollection 2015 Jan. PLoS Genet. 2015. PMID: 25569256 Free PMC article.
References
-
- Rubin-Bejerano I, Mandel S, Robzyk K, Kassir Y. Induction of meiosis in Saccharomyces cerevisiae depends on conversion of the transcriptional represssor Ume6 to a positive regulator by its regulated association with the transcriptional activator Ime1. Mol Cell Biol. 1996;16:2518–2526. - PMC - PubMed
-
- Covitz PA, Mitchell AP. Repression by the yeast meiotic inhibitor RME1. Genes Dev. 1993;7:1598–1608. - PubMed
-
- Sagee S, Sherman A, Shenhar G, Robzyk K, Ben-Doy N, Simchen G, Kassir Y. Multiple and distinct activation and repression sequences mediate the regulated transcription of IME1, a transcriptional activator of meiosis-specific genes in Saccharomyces cerevisiae. Mol Cell Biol. 1998;18:1985–1995. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

