Reduction in bone relapse and improved survival with oral clodronate for adjuvant treatment of operable breast cancer [ISRCTN83688026]
- PMID: 16542503
- PMCID: PMC1557723
- DOI: 10.1186/bcr1384
Reduction in bone relapse and improved survival with oral clodronate for adjuvant treatment of operable breast cancer [ISRCTN83688026]
Erratum in
- Breast Cancer Res. 2006;8(3):406
Abstract
Introduction: Experimental and clinical data show that the oral bisphosphonate clodronate (Bonefos) can inhibit tumor-induced osteoclastic bone resorption. This randomized, double-blind, placebo-controlled, multicenter trial was designed to determine if the addition of oral clodronate to standard treatment for primary operable breast cancer could reduce the subsequent occurrence of bone metastases and thereby improve overall survival.
Methods: 1,069 patients with primary operable stage I-III breast cancer were randomized to receive oral clodronate (1,600 mg/day) or placebo for 2 years, in conjunction with standard treatment for primary breast cancer including surgery, radiotherapy, adjuvant chemotherapy, and/or tamoxifen. All patients were assessed for bone metastases at two and five years and additionally when clinically indicated. Survival status was determined as of the close of the study on 30 June 2000 with a median follow up of 5.6 years. The treatment arms were compared using the unstratified log-rank test. Hazard ratios (HRs) with 95% confidence intervals were calculated.
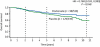
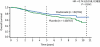
Results: Oral clodronate significantly reduced the risk of bone metastases in all patients over the 5 year study period (51 patients versus 73 patients with placebo; HR = 0.692, P = 0.043); the difference was also statistically significant over the 2 year medication period (19 patients versus 35 patients with placebo; HR = 0.546, P = 0.031). These differences were most pronounced in patients with stage II/III disease (39 patients versus 64 patients with placebo, HR = 0.592, P = 0.009 over 5 years; 16 patients versus 32 patients with placebo, HR= 0.496, P = 0.020 over 2 years). Survival data also favoured the clodronate arm (HR for all patients = 0.768, P = 0.048; HR for stage II/III disease = 0.743, P = 0.041), although this was not significant due to multiple analyses. Oral clodronate was well tolerated, with mild-to-moderate diarrhoea being the most frequently reported adverse event.
Conclusion: These results confirm that oral clodronate will significantly improve the 5 year bone relapse free survival when used as a supplementary adjuvant treatment for patients receiving standard treatment for primary operable breast cancer.
Figures



Similar articles
-
Randomized, placebo-controlled trial of clodronate in patients with primary operable breast cancer.J Clin Oncol. 2002 Aug 1;20(15):3219-24. doi: 10.1200/JCO.2002.11.080. J Clin Oncol. 2002. PMID: 12149294 Clinical Trial.
-
[Double-blinded controlled study comparing clodronate versus placebo in patients with breast cancer bone metastases].Bull Cancer. 2001 Jul;88(7):701-7. Bull Cancer. 2001. PMID: 11495824 Clinical Trial. French.
-
Oral bisphosphonates as adjuvant therapy for operable breast cancer.Clin Cancer Res. 2006 Oct 15;12(20 Pt 2):6301s-6304s. doi: 10.1158/1078-0432.CCR-06-1211. Clin Cancer Res. 2006. PMID: 17062718 Review.
-
Reduction in new metastases in breast cancer with adjuvant clodronate treatment.N Engl J Med. 1998 Aug 6;339(6):357-63. doi: 10.1056/NEJM199808063390601. N Engl J Med. 1998. PMID: 9691101 Clinical Trial.
-
Current and future status of adjuvant therapy for breast cancer.Cancer. 2003 Feb 1;97(3 Suppl):880-6. doi: 10.1002/cncr.11124. Cancer. 2003. PMID: 12548590 Review.
Cited by
-
Can Zoledronic Acid be Beneficial for Promoting Tumor Response in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy?J Clin Med. 2013 Oct 16;2(4):188-200. doi: 10.3390/jcm2040188. J Clin Med. 2013. PMID: 26237143 Free PMC article.
-
Cancer-associated bone disease.Curr Osteoporos Rep. 2007 Sep;5(3):120-7. doi: 10.1007/s11914-007-0027-8. Curr Osteoporos Rep. 2007. PMID: 17925193 Review.
-
Bisphosphonate and risk of cancer recurrence: protocol for a systematic review, meta-analysis and trial sequential analysis of randomised controlled trials.BMJ Open. 2015 Apr 21;5(4):e007215. doi: 10.1136/bmjopen-2014-007215. BMJ Open. 2015. PMID: 25900461 Free PMC article.
-
NCCN Task Force Report: Bone Health in Cancer Care.J Natl Compr Canc Netw. 2009 Jun;7 Suppl 3(Suppl 3):S1-32; quiz S33-5. doi: 10.6004/jnccn.2009.0076. J Natl Compr Canc Netw. 2009. PMID: 19555589 Free PMC article.
-
Treatment of osteoporosis and reduction in risk of invasive breast cancer in postmenopausal women with raloxifene.Expert Opin Pharmacother. 2011 Mar;12(4):657-74. doi: 10.1517/14656566.2011.557360. Epub 2011 Feb 7. Expert Opin Pharmacother. 2011. Retraction in: Expert Opin Pharmacother. 2012 May;13(7):1081. doi: 10.1517/14656566.2012.677336. PMID: 21294695 Free PMC article. Retracted. Review.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

