Therapeutic application of transcranial magnetic stimulation in Parkinson's disease: the contribution of expectation
- PMID: 16545582
- PMCID: PMC2967525
- DOI: 10.1016/j.neuroimage.2006.02.005
Therapeutic application of transcranial magnetic stimulation in Parkinson's disease: the contribution of expectation
Abstract
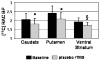
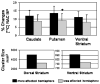
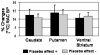
Repetitive transcranial magnetic stimulation (rTMS) is a valuable probe of brain function. Ever since its adoption as a research tool, there has been great interest regarding its potential clinical role. Presently, it is unclear whether rTMS will have some role as an alternative treatment for neuropsychiatric and neurological disorders such as Parkinson's disease (PD). To date, studies addressing the contribution of placebo during rTMS are missing. The placebo effect has been shown to be associated either with release of dopamine in the striatum or with changes in brain glucose metabolism. The main objective of this study was to test whether, in patients with PD, the expectation of therapeutic benefit from rTMS, which actually was delivered only as sham rTMS (placebo-rTMS) induced changes in striatal [11C] raclopride binding potentials (BP) as measured with positron emission tomography (PET). Placebo-rTMS induced a significant bilateral reduction in [11C] raclopride BP in dorsal and ventral striatum as compared to the baseline condition. This reduction BP is indicative of an increase in dopamine neurotransmission. The changes in [11C] raclopride binding were more evident in the hemisphere contralateral to the more affected side supporting the hypothesis that the more severe the symptoms, the greater the drive for symptom relief, and therefore the placebo response. This is the first study addressing the placebo contribution during rTMS. While our results seem to confirm earlier evidence that expectation induces dopaminergic placebo effects, they also suggest the importance of placebo-controlled studies for future clinical trials involving brain stimulation techniques.
Figures


References
-
- Aston JA, Gunn RN, Worsley KJ, Ma Y, Evans AC, Dagher A. A statistical method for the analysis of positron emission tomography neuroreceptor ligand data. NeuroImage. 2000;12:245–256. - PubMed
-
- Benedetti F, Colloca L, Torre E, Lanotte M, Melcarne A, Pesare M, Bergamasco B, Lopiano L. Placebo-responsive Parkinson patients show decreased activity in single neurons of subthalamic nucleus. Nat Neurosci. 2004;7 (6):587–588. - PubMed
-
- Breier A, Su TP, Saunders R, Carson RE, Kolachana BS, De Bartolomeis A, Weinberger DR, Weisenfeld N, Malhotra AK, Eckelman WC, Pickar D. Effects of NMDA antagonism on striatal dopamine release in healthy subjects: application of a novel PET approach. Proc Natl Acad Sci U S A. 1997;29:142–147. - PubMed
-
- Canavero S, Bonicalzi V, Paolotti R, Castellano G, Greco-Crasto S, Rizzo L, Davini O, Maina R. Therapeutic extradural cortical stimulation for movement disorders: a review. Neurol Res. 2003;25:118–122. - PubMed
-
- Collins DL, Neelin P, Peters TM, Evans AC. Automatic 3D intersubject registration of MR volumetric data in standardized Talairach space. J Comput Assist Tomogr. 1994;18:192–205. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

