Role of the property of C-reactive protein to activate the classical pathway of complement in protecting mice from pneumococcal infection
- PMID: 16547275
- PMCID: PMC3818093
- DOI: 10.4049/jimmunol.176.7.4369
Role of the property of C-reactive protein to activate the classical pathway of complement in protecting mice from pneumococcal infection
Abstract
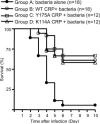
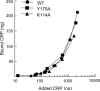
C-reactive protein (CRP) is not an acute-phase protein in mice, and therefore, mice are widely used to investigate the functions of human CRP. It has been shown that CRP protects mice from pneumococcal infection, and an active complement system is required for full protection. In this study, we assessed the contribution of CRP's ability of activating the classical pathway of complement in the protection of mice from lethal infection with virulent Streptococcus pneumoniae type 3. We used two CRP mutants, Y175A and K114A. The Y175A CRP does not bind C1q and does not activate complement in human serum. The K114A CRP binds C1q and activates complement more efficiently than wild-type CRP. Passively administered, both CRP mutants and the wild-type CRP protected mice from infection equally. Infected mice injected with wild-type or mutant CRP had reduced bacteremia, resulting in lower mortality and increased longevity compared with mice that did not receive CRP. Thus, the protection of mice was independent of CRP-mediated activation of the classical pathway of complement. To confirm that human CRP does not differentiate between human and mouse complement, we analyzed the binding of human CRP to mouse C1q. Surprisingly, CRP did not react with mouse C1q, although both mutant and wild-type CRP activated mouse C3, indicating species specificity of CRP-C1q interaction. We conclude that the mouse is an unfit animal for exploring CRP-mediated activation of the classical complement pathway, and that the characteristic of CRP to activate the classical complement pathway has no role in protecting mice from infection.
Figures
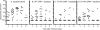
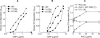
Similar articles
-
Human C-reactive protein protects mice from Streptococcus pneumoniae infection without binding to pneumococcal C-polysaccharide.J Immunol. 2007 Jan 15;178(2):1158-1163. doi: 10.4049/jimmunol.178.2.1158. J Immunol. 2007. PMID: 17202380 Free PMC article.
-
Protection from Streptococcus pneumoniae infection by C-reactive protein and natural antibody requires complement but not Fc gamma receptors.J Immunol. 2002 Jun 15;168(12):6375-81. doi: 10.4049/jimmunol.168.12.6375. J Immunol. 2002. PMID: 12055255
-
Complement Activation by C-Reactive Protein Is Critical for Protection of Mice Against Pneumococcal Infection.Front Immunol. 2020 Aug 13;11:1812. doi: 10.3389/fimmu.2020.01812. eCollection 2020. Front Immunol. 2020. PMID: 32903624 Free PMC article.
-
C-reactive protein reactivity with complement and effects on phagocytosis.Ann N Y Acad Sci. 1982;389:251-62. doi: 10.1111/j.1749-6632.1982.tb22141.x. Ann N Y Acad Sci. 1982. PMID: 7046579 Review.
-
The protective function of human C-reactive protein in mouse models of Streptococcus pneumoniae infection.Endocr Metab Immune Disord Drug Targets. 2008 Dec;8(4):231-7. doi: 10.2174/187153008786848321. Endocr Metab Immune Disord Drug Targets. 2008. PMID: 19075776 Free PMC article. Review.
Cited by
-
C-reactive protein collaborates with plasma lectins to boost immune response against bacteria.EMBO J. 2007 Jul 25;26(14):3431-40. doi: 10.1038/sj.emboj.7601762. Epub 2007 Jun 21. EMBO J. 2007. PMID: 17581635 Free PMC article.
-
Inflammation and Nutritional Science for Programs/Policies and Interpretation of Research Evidence (INSPIRE).J Nutr. 2015 May;145(5):1039S-1108S. doi: 10.3945/jn.114.194571. Epub 2015 Apr 1. J Nutr. 2015. PMID: 25833893 Free PMC article. Review.
-
Identification of Lipid Biomarkers for Chronic Joint Pain Associated with Different Joint Diseases.Biomolecules. 2023 Feb 9;13(2):342. doi: 10.3390/biom13020342. Biomolecules. 2023. PMID: 36830710 Free PMC article.
-
Age-Specific Differences in Inflammatory Biomarkers and Their Impact on Futile Recanalization After Mechanical Thrombectomy: An Inverse Probability Weighting Analysis.Eur J Neurol. 2025 May;32(5):e70182. doi: 10.1111/ene.70182. Eur J Neurol. 2025. PMID: 40353608 Free PMC article.
-
Evolution of C-Reactive Protein.Front Immunol. 2019 Apr 30;10:943. doi: 10.3389/fimmu.2019.00943. eCollection 2019. Front Immunol. 2019. PMID: 31114584 Free PMC article. Review.
References
-
- Abernethy TJ, Avery OT. The occurrence during acute infections of a protein not normally present in the blood. I. Distribution of the reactive protein in patients’ sera and the effect of calcium on the flocculation reaction with C polysaccharide of pneumococcus. J. Exp. Med. 1940;73:173–182. - PMC - PubMed
-
- Volanakis JE, Kaplan MH. Specificity of C-reactive protein for choline phosphate residues of pneumococcal C-polysaccharide. Proc. Soc. Exp. Biol. Med. 1971;136:612–614. - PubMed
-
- Agrawal A, Simpson MJ, Black S, Carey MP, Samols D. A C-reactive protein mutant that does not bind to phosphocholine and pneumococcal C-polysaccharide. J. Immunol. 2002;169:3217–3222. - PubMed
-
- Kaplan MH, Volanakis JE. Interaction of C-reactive protein complexes with the complement system. I. Consumption of human complement associated with the reaction of C-reactive protein with pneumococcal C-polysaccharide and with the choline phosphatides, lecithin, and sphingomyelin. J. Immunol. 1974;112:2135–2147. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

