Mechanoporation induced by diffuse traumatic brain injury: an irreversible or reversible response to injury?
- PMID: 16554464
- PMCID: PMC6674089
- DOI: 10.1523/JNEUROSCI.5119-05.2006
Mechanoporation induced by diffuse traumatic brain injury: an irreversible or reversible response to injury?
Abstract
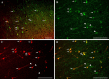
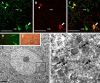
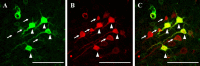
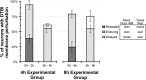
Diffuse traumatic brain injury (DTBI) is associated with neuronal plasmalemmal disruption, leading to either necrosis or reactive change without cell death. This study examined whether enduring membrane perturbation consistently occurs, leading to cell death, or if there is the potential for transient perturbation followed by resealing/recovery. We also examined the relationship of these events to calpain-mediated spectrin proteolysis (CMSP). To assess plasmalemmal disruption, rats (n = 21) received intracerebroventricular infusion 2 h before DTBI of a normally excluded 10 kDa fluorophore-labeled dextran. To reveal plasmalemmal resealing or enduring disruption, rats were infused with another labeled dextran 2 h (n = 10) or 6 h (n = 11) after injury. Immunohistochemistry for the 150 kDa spectrin breakdown product evaluated the concomitant role of CMSP. Neocortical neurons were followed with confocal and electron microscopy. After DTBI at 4 and 8 h, 55% of all tracer-flooded neurons contained both dextrans, demonstrating enduring plasmalemmal leakage, with many demonstrating necrosis. At 4 h, 12.0% and at 8 h, 15.7% of the dual tracer-flooded neurons showed CMSP, yet, these demonstrated less advanced cellular change. At 4 h, 39.0% and at 8 h, 24.4% of all tracer-flooded neurons revealed only preinjury dextran uptake, consistent with membrane resealing, whereas 7.6 and 11.1%, respectively, showed CMSP. At 4 h, 35% and at 8 h, 33% of neurons demonstrated CMSP without dextran flooding. At 4 h, 5.5% and at 8 h, 20.9% of tracer-flooded neurons revealed only postinjury dextran uptake, consistent with delayed membrane perturbation, with 55.0 and 35.4%, respectively, showing CMSP. These studies illustrate that DTBI evokes evolving plasmalemmal changes that highlight mechanical and potential secondary events in membrane poration.
Figures






References
-
- Amorini AM, Dunbar JG, Marmarou A (2003). Modulation of aquaporin-4 water transport in a model of TBI. Acta Neurochir Suppl 86:261–263. - PubMed
-
- Andrews NW (2005). Membrane resealing: synaptotagmin VII keeps running the show. Sci STKE 2005:e19. - PubMed
-
- Bramlett HM, Dietrich WD (2004). Pathophysiology of cerebral ischemia and brain trauma: similarities and differences. J Cereb Blood Flow Metab 24:133–150. - PubMed
-
- Brana C, Benham CD, Sundstrom LE (1999). Calpain activation and inhibition in organotypic rat hippocampal slice cultures deprived of oxygen and glucose. Eur J Neurosci 11:2375–2384. - PubMed
-
- Buki A, Siman R, Trojanowski JQ, Povlishock JT (1999). The role of calpain-mediated spectrin proteolysis in traumatically induced axonal injury. J Neuropathol Exp Neurol 58:365–375. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
