System Xc- and apolipoprotein E expressed by microglia have opposite effects on the neurotoxicity of amyloid-beta peptide 1-40
- PMID: 16554485
- PMCID: PMC6674113
- DOI: 10.1523/JNEUROSCI.5186-05.2006
System Xc- and apolipoprotein E expressed by microglia have opposite effects on the neurotoxicity of amyloid-beta peptide 1-40
Abstract
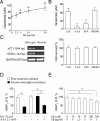
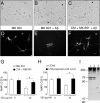
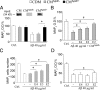
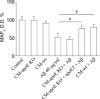
Because senile plaques in Alzheimer's disease (AD) contain reactive microglia in addition to potentially neurotoxic aggregates of amyloid-beta (Abeta), we examined the influence of microglia on the viability of rodent neurons in culture exposed to aggregated Abeta 1-40. Microglia enhanced the toxicity of Abeta by releasing glutamate through the cystine-glutamate antiporter system Xc-. This may be relevant to Abeta toxicity in AD, because the system Xc(-)-specific xCT gene is expressed not only in cultured microglia but also in reactive microglia within or surrounding amyloid plaques in transgenic mice expressing mutant human amyloid precursor protein or in wild-type mice injected with Abeta. Inhibition of NMDA receptors or system Xc- prevented the microglia-enhanced neurotoxicity of Abeta but also unmasked a neuroprotective effect of microglia mediated by microglial secretion of apolipoprotein E (apoE) in the culture medium. Immunodepletion of apoE or targeted inactivation of the apoE gene in microglia abrogated neuroprotection by microglial conditioned medium, whereas supplementation by human apoE isoforms restored protection, which was potentiated by the presence of microglia-derived cofactors. These results suggest that inhibition of microglial system Xc- might be of therapeutic value in the treatment of AD. Its inhibition not only prevents glutamate excitotoxicity but also facilitates neuroprotection by apoE.
Figures






References
-
- Bales KR, Verina T, Cummins DJ, Du Y, Dodel RC, Saura J, Fishman CE, DeLong CA, Piccardo P, Petegnief V, Ghetti B, Paul SM (1999). Apolipoprotein E is essential for amyloid deposition in the APP(V717F) transgenic mouse model of Alzheimer's disease. Proc Natl Acad Sci USA 96:15233–15238. - PMC - PubMed
-
- Bannai S, Sato H, Ishii T, Sugita Y (1989). Induction of cystine transport activity in human fibroblasts by oxygen. J Biol Chem 264:18480–18484. - PubMed
-
- Barger SW, Basile AS (2001). Activation of microglia by secreted amyloid precursor protein evokes release of glutamate by cystine exchange and attenuates synaptic function. J Neurochem 76:846–854. - PubMed
-
- Beutler HO (1985). l-Glutamate, colorimetric method with glutamate dehydrogenase and diaphorase. In: Methods of enzymatic analysis (Grassl M, ed.) pp. 369–376. Weinheim, Germany: VCH Verlagsgesellschaft mbH.
-
- Blanchard V, Moussaoui S, Czech C, Touchet N, Bonici B, Planche M, Canton T, Jedidi I, Gohin M, Wirths O, Bayer TA, Langui D, Duyckaerts C, Tremp G, Pradier L (2003). Time sequence of maturation of dystrophic neurites associated with Abeta deposits in APP/PS1 transgenic mice. Exp Neurol 184:247–263. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
