Contribution of rpoB2 RNA polymerase beta subunit gene to rifampin resistance in Nocardia species
- PMID: 16569850
- PMCID: PMC1426977
- DOI: 10.1128/AAC.50.4.1342-1346.2006
Contribution of rpoB2 RNA polymerase beta subunit gene to rifampin resistance in Nocardia species
Abstract
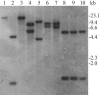
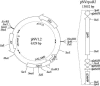
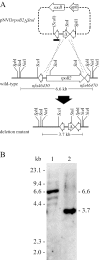
Nocardia species are gram-positive environmental saprophytes, but some cause the infectious disease nocardiosis. The complete genomic sequence of Nocardia farcinica IFM 10152 has been determined, and analyses indicated the presence of two different RNA polymerase beta subunit genes, rpoB and rpoB2, in the genome (J. Ishikawa, A. Yamashita, Y. Mikami, Y. Hoshino, H. Kurita, K. Hotta, T. Shiba, and M. Hattori, Proc. Natl. Acad. Sci. USA 101:14925-14930, 2004). These genes share 88.8% identity at the nucleotide level. Moreover, comparison of their amino acid sequences with those of other bacterial RpoB proteins suggested that the nocardial RpoB protein is likely to be rifampin (RIF) sensitive, whereas RpoB2 protein contains substitutions at the RIF-binding region that are likely to confer RIF resistance. Southern analysis indicated that rpoB duplication is widespread in Nocardia species and is correlated with the RIF-resistant phenotype. The introduction of rpoB2 by using a newly developed Nocardia-Escherichia coli shuttle plasmid vector and transformation system conferred RIF resistance to Nocardia asteroides IFM 0319T, which has neither RIF resistance nor rpoB duplication. Furthermore, unmarked rpoB2 deletion mutants of N. farcinica IFM 10152 showed no significant resistance to RIF. These results indicated the contribution of rpoB2 to RIF resistance in Nocardia species. Since this is the first example of genetic engineering of the Nocardia genome, we believe that this study, as well as our determination of the N. farcinica genome sequence, will be a landmark in Nocardia genetics.
Figures

Similar articles
-
Genetic bases of the rifampin resistance phenotype in Brucella spp.J Clin Microbiol. 2004 Dec;42(12):5439-43. doi: 10.1128/JCM.42.12.5439-5443.2004. J Clin Microbiol. 2004. PMID: 15583262 Free PMC article.
-
Monooxygenation of rifampicin catalyzed by the rox gene product of Nocardia farcinica: structure elucidation, gene identification and role in drug resistance.J Antibiot (Tokyo). 2010 Jan;63(1):23-8. doi: 10.1038/ja.2009.116. Epub 2009 Nov 27. J Antibiot (Tokyo). 2010. PMID: 19942945
-
Detection of rpoB mutations associated with rifampin resistance in Mycobacterium tuberculosis using denaturing gradient gel electrophoresis.Antimicrob Agents Chemother. 2005 Jun;49(6):2200-9. doi: 10.1128/AAC.49.6.2200-2209.2005. Antimicrob Agents Chemother. 2005. PMID: 15917513 Free PMC article.
-
[Recent progress in taxonomic studies on pathogenic nocardia and usefulness of the bacteria for the studies on secondary metabolites and antibiotic resistant mechanisms].Nihon Ishinkin Gakkai Zasshi. 2010;51(4):179-92. doi: 10.3314/jjmm.51.179. Nihon Ishinkin Gakkai Zasshi. 2010. PMID: 21060211 Review. Japanese.
-
Rifampicin-resistance, rpoB polymorphism and RNA polymerase genetic engineering.J Biotechnol. 2015 May 20;202:60-77. doi: 10.1016/j.jbiotec.2014.11.024. Epub 2014 Dec 3. J Biotechnol. 2015. PMID: 25481100 Review.
Cited by
-
Homozygous triplicate mutations in three 16S rRNA genes responsible for high-level aminoglycoside resistance in Nocardia farcinica clinical isolates from a Canada-wide bovine mastitis epizootic.Antimicrob Agents Chemother. 2010 Jun;54(6):2385-90. doi: 10.1128/AAC.00021-10. Epub 2010 Mar 22. Antimicrob Agents Chemother. 2010. PMID: 20308368 Free PMC article.
-
Nfa34810 Facilitates Nocardia farcinica Invasion of Host Cells and Stimulates Tumor Necrosis Factor Alpha Secretion through Activation of the NF-κB and Mitogen-Activated Protein Kinase Pathways via Toll-Like Receptor 4.Infect Immun. 2020 Mar 23;88(4):e00831-19. doi: 10.1128/IAI.00831-19. Print 2020 Mar 23. Infect Immun. 2020. PMID: 31964749 Free PMC article.
-
Reassessment of the genetic basis of natural rifampin resistance in the genus Rickettsia.Microbiologyopen. 2024 Aug;13(4):e1431. doi: 10.1002/mbo3.1431. Microbiologyopen. 2024. PMID: 39082505 Free PMC article.
-
Characterization of a rifampin-inactivating glycosyltransferase from a screen of environmental actinomycetes.Antimicrob Agents Chemother. 2012 Oct;56(10):5061-9. doi: 10.1128/AAC.01166-12. Epub 2012 Jul 16. Antimicrob Agents Chemother. 2012. PMID: 22802246 Free PMC article.
-
Identification of nocobactin NA biosynthetic gene clusters in Nocardia farcinica.J Bacteriol. 2011 Jan;193(2):441-8. doi: 10.1128/JB.00897-10. Epub 2010 Nov 19. J Bacteriol. 2011. PMID: 21097631 Free PMC article.
References
-
- Andressen, D. I., and B. R. Levin. 1999. Persistence of antibiotic resistant bacteria. Curr. Opin. Microbiol. 2:489-493. - PubMed
-
- Bibb, M. J., M. J. Bibb, J. M. Ward, and S. N. Cohen. 1985. Nucleotide sequences encoding and promoting expression of three antibiotic resistance genes indigenous to Streptomyces. Mol. Gen. Genet. 199:26-36. - PubMed
-
- Inaoka, T., K. Takahashi, H. Yada, M. Yoshida, and K. Ochi. 2004. RNA polymerase mutation activates the production of a dormant antibiotic 3,3′-neotrehalosadiamine via an autoinduction mechanism in Bacillus subtilis. J. Biol. Chem. 279:3885-3892. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

