Phase II study of erlotinib (OSI-774) in patients with metastatic colorectal cancer
- PMID: 16570047
- PMCID: PMC2361254
- DOI: 10.1038/sj.bjc.6603055
Phase II study of erlotinib (OSI-774) in patients with metastatic colorectal cancer
Abstract
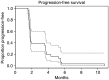
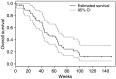
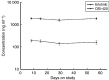
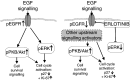
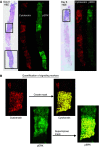
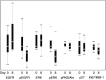
Erlotinib (Tarceva, OSI-774), a potent epidermal growth factor receptor tyrosine kinase inhibitor (EGFR), was evaluated in a phase II study to assess its activity in patients with metastatic colorectal cancer. In all, 38 patients with metastatic colorectal cancer were treated with erlotinib at a continuous daily oral dose of 150 mg. Radiological evaluation was carried out every 8 weeks and tumour biopsies were performed before treatment and on day 8. Of 31 evaluable patients, 19 (61%) had progressive disease and 12 (39%) had stable disease (s.d.). The median time to progression for those patients having s.d. was 123 days (range 108-329 days). The most common adverse events were rash in 34 patients and diarrhoea in 23 patients. Correlative studies were conducted to investigate the effect of erlotinib on downstream signalling. Tumour tissue correlations were based on usable tissue from eight match paired tumour samples pre- and on therapy, and showed a statistically significant decrease in the median intensity of both pEGFR (P=0.008) and phospho-extracellular signal-regulated kinase (ERK) (P=0.008) a week after commencement of treatment. No other statistically significant change in tumour markers was observed. Erlotinib was well tolerated with the most common toxicities being rash and diarrhoea. More than one-third of evaluable patients had s.d. for a minimum of 8 weeks. Correlative studies showed a reduction in phosphorylated EGFR and ERK in tumour tissue post-treatment.
Figures
References
-
- Carpenter G, Cohen S (1990) Epidermal growth factor. J Biol Chem 265: 7709–7712 - PubMed
-
- Cunningham D, Humblet Y, Siena S, Khayat D, Bleiberg H, Santoro A, Bets D, Mueser M, Harstrick A, Verslype C, Chau I, Van Cutsem E (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan refractory metastatic colorectal cancer. N Engl J Med 351: 337–345 - PubMed
-
- Fisher GA, Kuo T, Cho CD, Halsey J, Jambalos CN, Schwartz EJ, Robert RV, Advani RH, Wakelee HA, Sikic BI (2004) A phase II study of gefitinib in combination with FOLFOX-4 (IFOX) in patients with metastatic colorectal cancer. Proc Am Soc Clin Oncol 22: 14S (abstr. 187)
-
- Giacchetti S, Perpoint B, Zidani R, Le Bail N, Faggiuolo R, Focan C, Chollet P, Llory JF, Letourneau Y, Coudert B, Bertheaut-Cvitkovic F, Larregain-Fournier D, Le Rol A, Walter S, Adam R, Misset JL, Levi F (2000) Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil–leucovorin as first-line treatment of metastatic colorectal cancer. J Clin Oncol 18: 136–147 - PubMed
-
- Gordon AN, Finkler N, Edwards RP, Garcia AA, Crozier M, Irwin DH, Barrett E (2005) Phase 2 evaluation of OSI-774, a potent oral antagonist of the EGFR-TK in patients with advanced ovarian cancer. Int J Gynecol Cancer 15: 785–792 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

