Persistent sodium current in layer 5 neocortical neurons is primarily generated in the proximal axon
- PMID: 16571753
- PMCID: PMC6673860
- DOI: 10.1523/JNEUROSCI.4907-05.2006
Persistent sodium current in layer 5 neocortical neurons is primarily generated in the proximal axon
Abstract
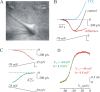
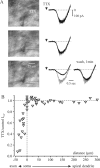
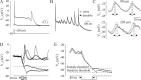
In addition to the well described fast-inactivating component of the Na+ current [transient Na+ current (INaT)], neocortical neurons also exhibit a low-voltage-activated, slowly inactivating "persistent" Na+ current (INaP), which plays a role in determining neuronal excitability and synaptic integration. We investigated the Na+ channels responsible for INaP in layer 5 pyramidal cells using cell-attached and whole-cell recordings in neocortical slices. In simultaneous cell-attached and whole-cell somatic recordings, no persistent Na+ channel activity was detected at potentials at which whole-cell INaP operates. Detailed kinetic analysis of late Na+ channel activity in cell-attached patches at 36 degrees C revealed that somatic Na+ channels do not demonstrate "modal gating" behavior and that the probability of single late openings is extremely low (<1.4 x 10(-4) or <0.02% of maximal open probability of INaT). Ensemble averages of these currents did not reveal a sustained component whose amplitude and voltage dependence could account for INaP as seen in whole-cell recordings. Local application of TTX to the axon blocked somatically recorded INaP, whereas somatic and dendritic application had little or no effect. Finally, simultaneous current-clamp recordings from soma and apical dendrite revealed that Na+ plateau potentials originate closer to the axon. Our data indicate that the primary source of INaP is in the spike initiation zone in the proximal axon. The focal axonal presence of regenerative subthreshold conductance with voltage and time dependence optimal to manipulate integration of synaptic input, spike threshold, and the pattern of repetitive firing provides the layer 5 pyramidal neuron with a mechanism for dynamic control of its gain.
Figures



Similar articles
-
Action potential initiation and propagation in rat neocortical pyramidal neurons.J Physiol. 1997 Dec 15;505 ( Pt 3)(Pt 3):617-32. doi: 10.1111/j.1469-7793.1997.617ba.x. J Physiol. 1997. PMID: 9457640 Free PMC article.
-
Dendritic Na+ channels amplify EPSPs in hippocampal CA1 pyramidal cells.J Neurophysiol. 1996 Oct;76(4):2181-91. doi: 10.1152/jn.1996.76.4.2181. J Neurophysiol. 1996. PMID: 8899593
-
Activation of protein kinase C increases neuronal excitability by regulating persistent Na+ current in mouse neocortical slices.J Neurophysiol. 1998 Sep;80(3):1547-51. doi: 10.1152/jn.1998.80.3.1547. J Neurophysiol. 1998. PMID: 9744958
-
Neuronal signaling in central nervous system.Sheng Li Xue Bao. 2011 Feb 25;63(1):1-8. Sheng Li Xue Bao. 2011. PMID: 21340428 Review.
-
Enhanced transmission of glutamate current flowing from the dendrite to the soma in rat neocortical layer 5 neurons.Novartis Found Symp. 2002;241:61-8; discussion 68-71, 226-32. Novartis Found Symp. 2002. PMID: 11771651 Review.
Cited by
-
Persistent Nav1.6 current at axon initial segments tunes spike timing of cerebellar granule cells.J Physiol. 2010 Feb 15;588(Pt 4):651-70. doi: 10.1113/jphysiol.2010.183798. J Physiol. 2010. PMID: 20173079 Free PMC article.
-
The role of spiking and bursting pacemakers in the neuronal control of breathing.J Biol Phys. 2011 Jun;37(3):241-61. doi: 10.1007/s10867-011-9214-z. Epub 2011 Mar 22. J Biol Phys. 2011. PMID: 22654176 Free PMC article.
-
Axon initial segment structure and function in health and disease.Physiol Rev. 2025 Apr 1;105(2):765-801. doi: 10.1152/physrev.00030.2024. Epub 2024 Oct 31. Physiol Rev. 2025. PMID: 39480263 Free PMC article. Review.
-
Single-Molecule Imaging of Nav1.6 on the Surface of Hippocampal Neurons Reveals Somatic Nanoclusters.Biophys J. 2016 Sep 20;111(6):1235-1247. doi: 10.1016/j.bpj.2016.08.016. Biophys J. 2016. PMID: 27653482 Free PMC article.
-
Endogenous polyamines regulate cortical neuronal excitability by blocking voltage-gated Na+ channels.Proc Natl Acad Sci U S A. 2008 Dec 2;105(48):18994-9. doi: 10.1073/pnas.0803464105. Epub 2008 Nov 19. Proc Natl Acad Sci U S A. 2008. PMID: 19020082 Free PMC article.
References
-
- Alzheimer C, Schwindt PC, Crill WE (1993a). Postnatal development of a persistent Na+ current in pyramidal neurons from rat sensorimotor cortex. J Neurophysiol 69:290–292. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
