Evidence for a functional link between uncoating of the human immunodeficiency virus type 1 core and nuclear import of the viral preintegration complex
- PMID: 16571788
- PMCID: PMC1440469
- DOI: 10.1128/JVI.80.8.3712-3720.2006
Evidence for a functional link between uncoating of the human immunodeficiency virus type 1 core and nuclear import of the viral preintegration complex
Abstract
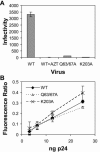
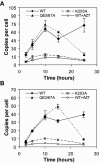
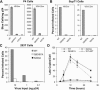
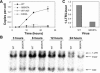
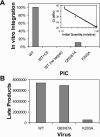
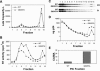
Human immunodeficiency virus type 1 (HIV-1) particles begin their replication upon fusion with the plasma membrane of target cells and release of the viral core into the host cell cytoplasm. Soon thereafter, the viral capsid, which is composed of a polymer of the CA protein, disassociates from the internal ribonucleoprotein complex. While this disassembly process remains poorly understood, the available evidence indicates that proper uncoating of the core is a key step in infection. Defects in uncoating most often lead to a failure of the virus to undergo reverse transcription, resulting in an inability to form a functional viral preintegration complex (PIC). In a previous study, we reported that an HIV-1 mutant containing two substitutions in CA (Q63A/67A) was unusual in that it was poorly infectious yet synthesized normal levels of viral DNA. Here we report that this mutant is impaired for nuclear entry. Quantitative analysis of viral DNA synthesis from infected cells by Southern blotting and real-time PCR revealed that the Q63A/Q67A mutant is impaired in the synthesis of one-long terminal repeat (1-LTR) and 2-LTR circles. Isolation of PICs from acutely infected cells revealed that the Q63A/Q67A mutant produces protein-DNA complexes similar to wild-type in yield and overall composition, but these PICs contained elevated levels of CA and were impaired for integration in vitro. These results demonstrate that mutations in CA can have deleterious effects on both nuclear targeting and integration, suggesting that these steps in the HIV-1 life cycle are dependent on proper uncoating of the viral core.
Figures

References
-
- Bouyac-Bertoia, M., J. D. Dvorin, R. A. Fouchier, Y. Jenkins, B. E. Meyer, L. I. Wu, M. Emerman, and M. H. Malim. 2001. HIV-1 infection requires a functional integrase NLS. Mol. Cell 7:1025-1035. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

