Analysis of rubella virus capsid protein-mediated enhancement of replicon replication and mutant rescue
- PMID: 16571813
- PMCID: PMC1440428
- DOI: 10.1128/JVI.80.8.3966-3974.2006
Analysis of rubella virus capsid protein-mediated enhancement of replicon replication and mutant rescue
Abstract
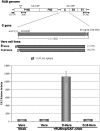
The rubella virus capsid protein (C) has been shown to complement a lethal deletion (termed deltaNotI) in P150 replicase protein. To investigate this phenomenon, we generated two lines of Vero cells that stably expressed either C (C-Vero cells) or C lacking the eight N-terminal residues (Cdelta8-Vero cells), a construct previously shown to be unable to complement DeltaNotI. In C-Vero cells but not Vero or Cdelta8-Vero cells, replication of a wild-type (wt) replicon expressing the green fluorescent protein (GFP) reporter gene (RUBrep/GFP) was enhanced, and replication of a replicon with deltaNotI (RUBrep/GFP-deltaNotI) was rescued. Surprisingly, replicons with deleterious mutations in the 5' and 3' cis-acting elements were also rescued in C-Vero cells. Interestingly, the Cdelta8 construct localized to the nucleus while the C construct localized in the cytoplasm, explaining the lack of enhancement and rescue in Cdelta8-Vero cells since rubella virus replication occurs in the cytoplasm. Enhancement and rescue in C-Vero cells were at a basic step in the replication cycle, resulting in a substantial increase in the accumulation of replicon-specific RNAs. There was no difference in translation of the nonstructural proteins in C-Vero and Vero cells transfected with the wt and mutant replicons, demonstrating that enhancement and rescue were not due to an increase in the efficiency of translation of the transfected replicon transcripts. In replicon-transfected C-Vero cells, C and the P150 replicase protein associated by coimmunoprecipitation, suggesting that C might play a role in RNA replication, which could explain the enhancement and rescue phenomena. A unifying model that accounts for enhancement of wt replicon replication and rescue of diverse mutations by the rubella virus C protein is proposed.
Figures


Similar articles
-
Rubella virus DI RNAs and replicons: requirement for nonstructural proteins acting in cis for amplification by helper virus.Virology. 2001 Oct 10;289(1):63-73. doi: 10.1006/viro.2001.1088. Virology. 2001. PMID: 11601918
-
Complementation of a deletion in the rubella virus p150 nonstructural protein by the viral capsid protein.J Virol. 2003 Sep;77(17):9502-10. doi: 10.1128/jvi.77.17.9502-9510.2003. J Virol. 2003. PMID: 12915564 Free PMC article.
-
Analysis of the 3' cis-acting elements of rubella virus by using replicons expressing a puromycin resistance gene.J Virol. 2004 Mar;78(5):2553-61. doi: 10.1128/jvi.78.5.2553-2561.2004. J Virol. 2004. PMID: 14963158 Free PMC article.
-
Characterization of cell lines stably transfected with rubella virus replicons.Virology. 2012 Jul 20;429(1):29-36. doi: 10.1016/j.virol.2012.04.003. Epub 2012 Apr 26. Virology. 2012. PMID: 22542003
-
Rubella virus capsid protein modulates viral genome replication and virus infectivity.J Virol. 2004 Apr;78(8):4314-22. doi: 10.1128/jvi.78.8.4314-4322.2004. J Virol. 2004. PMID: 15047844 Free PMC article.
Cited by
-
Rubella virus-like replicon particles: analysis of encapsidation determinants and non-structural roles of capsid protein in early post-entry replication.J Gen Virol. 2012 Mar;93(Pt 3):516-525. doi: 10.1099/vir.0.038984-0. Epub 2011 Nov 23. J Gen Virol. 2012. PMID: 22113006 Free PMC article.
-
Determinants in the maturation of rubella virus p200 replicase polyprotein precursor.J Virol. 2012 Jun;86(12):6457-69. doi: 10.1128/JVI.06132-11. Epub 2012 Apr 4. J Virol. 2012. PMID: 22491463 Free PMC article.
-
Short self-interacting N-terminal region of rubella virus capsid protein is essential for cooperative actions of capsid and nonstructural p150 proteins.J Virol. 2014 Oct;88(19):11187-98. doi: 10.1128/JVI.01758-14. Epub 2014 Jul 23. J Virol. 2014. PMID: 25056903 Free PMC article.
-
The hepatitis C virus core protein can modulate RNA-dependent RNA synthesis by the 2a polymerase.Virus Res. 2014 Aug 30;189:165-76. doi: 10.1016/j.virusres.2014.05.017. Epub 2014 May 26. Virus Res. 2014. PMID: 24874198 Free PMC article.
-
Specific, sensitive, high-resolution detection of protein molecules in eukaryotic cells using metal-tagging transmission electron microscopy.Structure. 2012 May 9;20(5):759-66. doi: 10.1016/j.str.2012.04.001. Structure. 2012. PMID: 22579245 Free PMC article.
References
-
- Baron, M. D., and K. Forsell. 1991. Oligomerization of the structural proteins of rubella virus. Virology 185:811-819. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

