The products of the herpes simplex virus type 1 immediate-early US1/US1.5 genes downregulate levels of S-phase-specific cyclins and facilitate virus replication in S-phase Vero cells
- PMID: 16571817
- PMCID: PMC1440436
- DOI: 10.1128/JVI.80.8.4005-4016.2006
The products of the herpes simplex virus type 1 immediate-early US1/US1.5 genes downregulate levels of S-phase-specific cyclins and facilitate virus replication in S-phase Vero cells
Abstract
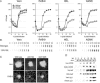
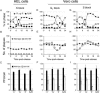
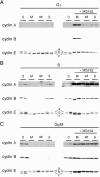
Herpes simplex virus type 1 ICP22-/U(S)1.5- mutants initiate viral gene expression in all cells; however, in most cell types, the replication process stalls due to an inability to express gamma2 late proteins. Although the function of ICP22/U(S)1.5 has not been established, it has been suggested that these proteins activate, induce, or repress the activity of cellular proteins during infection. In this study, we hypothesized that cell cycle-associated proteins are targets of ICP22/U(S)1.5. For this purpose, we first isolated and characterized an ICP22-/U(S)1.5- mutant virus, 22/n199. Like other ICP22-/U(S)1.5- mutants, 22/n199 replicates in a cell-type-specific manner and fails to induce efficient gamma2 late gene expression in restrictive cells. Although synchronization of restrictive human embryonic lung cells in each phase of the cell cycle did not overcome the growth restrictions of 22/n199, synchronization of permissive Vero cells in S phase rendered them less able to support 22/n199 plaque formation and replication. Consistent with this finding, expression of cellular S-phase cyclins was altered in an ICP22/U(S)1.5-dependent manner specifically when S-phase Vero cells were infected. Collectively, these observations support the notion that ICP22/U(S)1.5 deregulates the cell cycle upon infection of S-phase permissive cells by altering expression of key cell cycle regulatory proteins either directly or indirectly.
Figures

Similar articles
-
ICP22 is required for wild-type composition and infectivity of herpes simplex virus type 1 virions.J Virol. 2006 Oct;80(19):9381-90. doi: 10.1128/JVI.01061-06. J Virol. 2006. PMID: 16973544 Free PMC article.
-
Characterization of regulatory functions of the HSV-1 immediate-early protein ICP22.Virology. 1996 Dec 15;226(2):393-402. doi: 10.1006/viro.1996.0667. Virology. 1996. PMID: 8955059
-
Structural and functional characterization of herpes simplex virus 1 immediate-early protein infected-cell protein 22.J Biochem. 2006 Jul;140(1):67-73. doi: 10.1093/jb/mvj135. J Biochem. 2006. PMID: 16877770
-
The cell cycle, cyclin-dependent kinases, and viral infections: new horizons and unexpected connections.Prog Cell Cycle Res. 2003;5:103-24. Prog Cell Cycle Res. 2003. PMID: 14593705 Review.
-
The herpes simplex virus type 1 infected cell protein 22.Virol Sin. 2010 Feb;25(1):1-7. doi: 10.1007/s12250-010-3080-x. Epub 2010 Feb 12. Virol Sin. 2010. PMID: 20960278 Free PMC article. Review.
Cited by
-
Persistent inflammation and neuronal loss in the mouse brain induced by a modified form of attenuated herpes simplex virus type I.Virol Sin. 2023 Feb;38(1):108-118. doi: 10.1016/j.virs.2022.11.008. Epub 2022 Nov 24. Virol Sin. 2023. PMID: 36436797 Free PMC article.
-
A Revision of Herpes Simplex Virus Type 1 Transcription: First, Repress; Then, Express.Microorganisms. 2024 Jan 26;12(2):262. doi: 10.3390/microorganisms12020262. Microorganisms. 2024. PMID: 38399666 Free PMC article. Review.
-
Transient expression of herpes simplex virus type 1 ICP22 represses viral promoter activity and complements the replication of an ICP22 null virus.J Virol. 2009 Sep;83(17):8733-43. doi: 10.1128/JVI.00810-09. Epub 2009 Jun 17. J Virol. 2009. PMID: 19535441 Free PMC article.
-
Herpes simplex virus immediate-early protein ICP22 triggers loss of serine 2-phosphorylated RNA polymerase II.J Virol. 2007 May;81(10):5091-101. doi: 10.1128/JVI.00184-07. Epub 2007 Mar 7. J Virol. 2007. PMID: 17344289 Free PMC article.
-
ICP22 is required for wild-type composition and infectivity of herpes simplex virus type 1 virions.J Virol. 2006 Oct;80(19):9381-90. doi: 10.1128/JVI.01061-06. J Virol. 2006. PMID: 16973544 Free PMC article.
References
-
- Advani, S. J., R. Brandimarti, R. R. Weichselbaum, and B. Roizman. 2000. The disappearance of cyclins A and B and the increase in activity of the G2/M-phase cellular kinase cdc2 in herpes simplex virus 1-infected cells require expression of the α22/US1.5 and UL13 viral genes. J. Virol. 74:8-15. - PMC - PubMed
-
- Aleem, E., H. Kiyokawa, and P. Kaldis. 2005. Cdc2-cyclin E complexes regulate the G1/S phase transition. Nat. Cell Biol. 7:831-836. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

