Cell cycle perturbations induced by infection with the coronavirus infectious bronchitis virus and their effect on virus replication
- PMID: 16571830
- PMCID: PMC1440480
- DOI: 10.1128/JVI.80.8.4147-4156.2006
Cell cycle perturbations induced by infection with the coronavirus infectious bronchitis virus and their effect on virus replication
Abstract
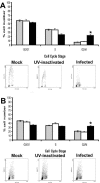
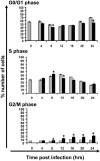
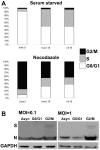
In eukaryotic cells, cell growth and division occur in a stepwise, orderly fashion described by a process known as the cell cycle. The relationship between positive-strand RNA viruses and the cell cycle and the concomitant effects on virus replication are not clearly understood. We have shown that infection of asynchronously replicating and synchronized replicating cells with the avian coronavirus infectious bronchitis virus (IBV), a positive-strand RNA virus, resulted in the accumulation of infected cells in the G2/M phase of the cell cycle. Analysis of various cell cycle-regulatory proteins and cellular morphology indicated that there was a down-regulation of cyclins D1 and D2 (G1 regulatory cyclins) and that a proportion of virus-infected cells underwent aberrant cytokinesis, in which the cells underwent nuclear, but not cytoplasmic, division. We assessed the impact of the perturbations on the cell cycle for virus-infected cells and found that IBV-infected G2/M-phase-synchronized cells exhibited increased viral protein production when released from the block when compared to cells synchronized in the G0 phase or asynchronously replicating cells. Our data suggested that IBV induces a G2/M phase arrest in infected cells to promote favorable conditions for viral replication.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

