Role of endosomal cathepsins in entry mediated by the Ebola virus glycoprotein
- PMID: 16571833
- PMCID: PMC1440424
- DOI: 10.1128/JVI.80.8.4174-4178.2006
Role of endosomal cathepsins in entry mediated by the Ebola virus glycoprotein
Abstract
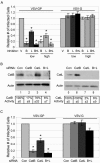
Using chemical inhibitors and small interfering RNA (siRNA), we have confirmed roles for cathepsin B (CatB) and cathepsin L (CatL) in Ebola virus glycoprotein (GP)-mediated infection. Treatment of Ebola virus GP pseudovirions with CatB and CatL converts GP1 from a 130-kDa to a 19-kDa species. Virus with 19-kDa GP1 displays significantly enhanced infection and is largely resistant to the effects of the CatB inhibitor and siRNA, but it still requires a low-pH-dependent endosomal/lysosomal function. These and other results support a model in which CatB and CatL prime GP by generating a 19-kDa intermediate that can be acted upon by an as yet unidentified endosomal/lysosomal enzyme to trigger fusion.
Figures
References
-
- Ebert, D. H., J. Deussing, C. Peters, and T. S. Dermody. 2002. Cathepsin L and cathepsin B mediate reovirus disassembly in murine fibroblast cells. J. Biol. Chem. 277:24609-24617. - PubMed
-
- Golden, J., and L. Schiff. 2005. Neutrophil elastase, an acid-independent serine protease, facilitates reovirus uncoating and infection in U937 promonocyte cells. Virol. J. 2:48. http://www.virologyj.com/content/2/1/48. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

