Cloned ferrets produced by somatic cell nuclear transfer
- PMID: 16584722
- PMCID: PMC1892907
- DOI: 10.1016/j.ydbio.2006.02.016
Cloned ferrets produced by somatic cell nuclear transfer
Abstract
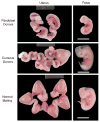
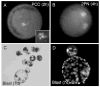
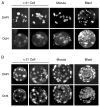
Somatic cell nuclear transfer (SCNT) offers great potential for developing better animal models of human disease. The domestic ferret (Mustela putorius furo) is an ideal animal model for influenza infections and potentially other human respiratory diseases such as cystic fibrosis, where mouse models have failed to reproduce the human disease phenotype. Here, we report the successful production of live cloned, reproductively competent, ferrets using species-specific SCNT methodologies. Critical to developing a successful SCNT protocol for the ferret was the finding that hormonal treatment, normally used for superovulation, adversely affected the developmental potential of recipient oocytes. The onset of Oct4 expression was delayed and incomplete in parthenogenetically activated oocytes collected from hormone-treated females relative to oocytes collected from females naturally mated with vasectomized males. Stimulation induced by mating and in vitro oocyte maturation produced the optimal oocyte recipient for SCNT. Although nuclear injection and cell fusion produced mid-term fetuses at equivalent rates (approximately 3-4%), only cell fusion gave rise to healthy surviving clones. Single cell fusion rates and the efficiency of SCNT were also enhanced by placing two somatic cells into the perivitelline space. These species-specific modifications facilitated the birth of live, healthy, and fertile cloned ferrets. The development of microsatellite genotyping for domestic ferrets confirmed that ferret clones were genetically derived from their respective somatic cells and unrelated to their surrogate mother. With this technology, it is now feasible to begin generating genetically defined ferrets for studying transmissible and inherited human lung diseases. Cloning of the domestic ferret may also aid in recovery and conservation of the endangered black-footed ferret and European mink.
Figures

Similar articles
-
Factors affecting the electrofusion of mouse and ferret oocytes with ferret somatic cells.Mol Reprod Dev. 2005 Sep;72(1):40-7. doi: 10.1002/mrd.20321. Mol Reprod Dev. 2005. PMID: 15948164
-
Factors affecting the efficiency of embryo transfer in the domestic ferret (Mustela putorius furo).Theriogenology. 2006 Jul 15;66(2):183-90. doi: 10.1016/j.theriogenology.2005.10.021. Epub 2005 Dec 5. Theriogenology. 2006. PMID: 16330092 Free PMC article.
-
Production of nuclear transfer-derived piglets using porcine fetal fibroblasts transfected with the enhanced green fluorescent protein.Biol Reprod. 2003 Sep;69(3):1060-8. doi: 10.1095/biolreprod.102.014886. Epub 2003 May 28. Biol Reprod. 2003. PMID: 12773429
-
Progress toward generating a ferret model of cystic fibrosis by somatic cell nuclear transfer.Reprod Biol Endocrinol. 2003 Nov 7;1:83. doi: 10.1186/1477-7827-1-83. Reprod Biol Endocrinol. 2003. PMID: 14613541 Free PMC article. Review.
-
Nuclear transfer in cats and its application.Theriogenology. 2006 Jul 1;66(1):72-81. doi: 10.1016/j.theriogenology.2006.03.017. Epub 2006 Apr 18. Theriogenology. 2006. PMID: 16620927 Review.
Cited by
-
Evolutionary perspectives on clonal reproduction in vertebrate animals.Proc Natl Acad Sci U S A. 2015 Jul 21;112(29):8867-73. doi: 10.1073/pnas.1501820112. Epub 2015 Jul 20. Proc Natl Acad Sci U S A. 2015. PMID: 26195735 Free PMC article.
-
Nuclear reprogramming of sperm and somatic nuclei in eggs and oocytes.Reprod Med Biol. 2013;12(4):133-149. doi: 10.1007/s12522-013-0155-z. Epub 2013 Jun 4. Reprod Med Biol. 2013. PMID: 24273450 Free PMC article.
-
Towards Practical Conservation Cloning: Understanding the Dichotomy Between the Histories of Commercial and Conservation Cloning.Animals (Basel). 2025 Mar 29;15(7):989. doi: 10.3390/ani15070989. Animals (Basel). 2025. PMID: 40218382 Free PMC article. Review.
-
Efficient term development of vitrified ferret embryos using a novel pipette chamber technique.Biol Reprod. 2008 Nov;79(5):832-40. doi: 10.1095/biolreprod.107.067371. Epub 2008 Jul 16. Biol Reprod. 2008. PMID: 18633142 Free PMC article.
-
Generation of SV40-transformed rabbit tracheal-epithelial-cell-derived blastocyst by somatic cell nuclear transfer.Cell Tissue Res. 2012 Feb;347(2):357-67. doi: 10.1007/s00441-011-1296-1. Epub 2012 Jan 12. Cell Tissue Res. 2012. PMID: 22234514 Free PMC article.
References
-
- Baguisi A, Behboodi E, Melican DT, Pollock JS, Destrempes MM, Cammuso C, Williams JL, Nims SD, Porter CA, Midura P, Palacios MJ, Ayres SL, Denniston RS, Hayes ML, Ziomek CA, Meade HM, Godke RA, Gavin WG, Overstrom EW, Echelard Y. Production of goats by somatic cell nuclear transfer. Nat Biotechnol. 1999;17:456–461. - PubMed
-
- Boquest AC, Grupen CG, Harrison SJ, McIlfatrick SM, Ashman RJ, d’Apice AJ, Nottle MB. Production of cloned pigs from cultured fetal fibroblast cells. Biol Reprod. 2002;66:1283–1287. - PubMed
-
- Bortvin A, Eggan K, Skaletsky H, Akutsu H, Berry DL, Yanagimachi R, Page DC, Jaenisch R. Incomplete reactivation of Oct4-related genes in mouse embryos cloned from somatic nuclei. Development. 2003;130:1673–1680. - PubMed
-
- Carter DB, Lai L, Park KW, Samuel M, Lattimer JC, Jordan KR, Estes DM, Besch-Williford C, Prather RS. Phenotyping of transgenic cloned piglets. Cloning Stem Cells. 2002;4:131–145. - PubMed
-
- Chang MC, Yanagimachi R. Fertilization of ferret ova by deposition of epididymal sperm into the ovarian capsule with special reference to the fertilizable lie of ova and the capacitation of sperm. J Exp Zool. 1963;154:175–187. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

