Green fluorescent chimeras indicate nonpolar localization of pullulanase secreton components PulL and PulM
- PMID: 16585754
- PMCID: PMC1447010
- DOI: 10.1128/JB.188.8.2928-2935.2006
Green fluorescent chimeras indicate nonpolar localization of pullulanase secreton components PulL and PulM
Abstract
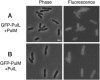
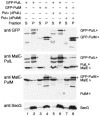
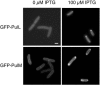
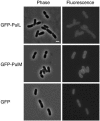
The Klebsiella oxytoca pullulanase secreton (type II secretion system) components PulM and PulL were tagged at their N termini with green fluorescent protein (GFP), and their subcellular location was examined by fluorescence microscopy and fractionation. When produced at moderate levels without other secreton components in Escherichia coli, both chimeras were envelope associated, as are the native proteins. Fluorescent GFP-PulM was evenly distributed over the cell envelope, with occasional brighter foci. Under the same conditions, GFP-PulL was barely detectable in the envelope by fluorescence microscopy. When produced together with all other secreton components, GFP-PulL exhibited circumferential fluorescence, with numerous brighter patches. The envelope-associated fluorescence of GFP-PulL was almost completely abolished when native PulL was also produced, suggesting that the chimera cannot compete with PulL for association with other secreton components. The patches of GFP-PulL might represent functional secretons, since GFP-PulM also appeared in similar patches. GFP-PulM and GFP-PulL both appeared in spherical polar foci when made at high levels. In K. oxytoca, GFP-PulM was evenly distributed over the cell envelope, with few patches, whereas GFP-PulL showed only weak envelope-associated fluorescence. These data suggest that, in contrast to their Vibrio cholerae Eps secreton counterparts (M. Scott, Z. Dossani, and M. Sandkvist, Proc. Natl. Acad. Sci. USA 98:13978-13983, 2001), PulM and PulL do not localize specifically to the cell poles and that the Pul secreton is distributed over the cell surface.
Figures

Similar articles
-
Multiple interactions between pullulanase secreton components involved in stabilization and cytoplasmic membrane association of PulE.J Bacteriol. 2000 Apr;182(8):2142-52. doi: 10.1128/JB.182.8.2142-2152.2000. J Bacteriol. 2000. PMID: 10735856 Free PMC article.
-
Type IV-like pili formed by the type II secreton: specificity, composition, bundling, polar localization, and surface presentation of peptides.J Bacteriol. 2003 Jun;185(11):3416-28. doi: 10.1128/JB.185.11.3416-3428.2003. J Bacteriol. 2003. PMID: 12754241 Free PMC article.
-
Disulfide bond formation in secreton component PulK provides a possible explanation for the role of DsbA in pullulanase secretion.J Bacteriol. 2001 Feb;183(4):1312-9. doi: 10.1128/JB.183.4.1312-1319.2001. J Bacteriol. 2001. PMID: 11157944 Free PMC article.
-
Macromolecular assembly and secretion across the bacterial cell envelope: type II protein secretion systems.J Mol Biol. 1998 Jun 12;279(3):485-99. doi: 10.1006/jmbi.1998.1791. J Mol Biol. 1998. PMID: 9641973 Review.
-
Green fluorescent protein as a reporter for macromolecular localization in bacterial cells.Methods. 2000 Jan;20(1):62-72. doi: 10.1006/meth.1999.0906. Methods. 2000. PMID: 10610805 Review.
Cited by
-
Docking and assembly of the type II secretion complex of Vibrio cholerae.J Bacteriol. 2009 May;191(9):3149-61. doi: 10.1128/JB.01701-08. Epub 2009 Feb 27. J Bacteriol. 2009. PMID: 19251862 Free PMC article.
-
The Apoptogenic Toxin AIP56 Is Secreted by the Type II Secretion System of Photobacterium damselae subsp. piscicida.Toxins (Basel). 2017 Nov 14;9(11):368. doi: 10.3390/toxins9110368. Toxins (Basel). 2017. PMID: 29135911 Free PMC article.
-
Signal recognition particle-dependent inner membrane targeting of the PulG Pseudopilin component of a type II secretion system.J Bacteriol. 2007 Mar;189(5):1783-93. doi: 10.1128/JB.01230-06. Epub 2006 Dec 8. J Bacteriol. 2007. PMID: 17158657 Free PMC article.
-
Artificial septal targeting of Bacillus subtilis cell division proteins in Escherichia coli: an interspecies approach to the study of protein-protein interactions in multiprotein complexes.J Bacteriol. 2008 Sep;190(18):6048-59. doi: 10.1128/JB.00462-08. Epub 2008 Jul 11. J Bacteriol. 2008. PMID: 18621900 Free PMC article.
-
FimL regulates cAMP synthesis in Pseudomonas aeruginosa.PLoS One. 2011 Jan 11;6(1):e15867. doi: 10.1371/journal.pone.0015867. PLoS One. 2011. PMID: 21264306 Free PMC article.
References
-
- Brandon, L. D., N. Goehring, A. Janakiraman, A. W. Yan, T. Wu, J. Beckwith, and M. B. Goldberg. 2003. IcsA, a polarly localized autotransporter with an atypical signal peptide, uses the Sec apparatus for secretion, although the Sec apparatus is circumferentially distributed. Mol. Microbiol. 50:45-60. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

