Correlation-induced synchronization of oscillations in olfactory bulb neurons
- PMID: 16597718
- PMCID: PMC6674124
- DOI: 10.1523/JNEUROSCI.4605-05.2006
Correlation-induced synchronization of oscillations in olfactory bulb neurons
Abstract
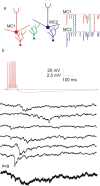
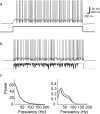
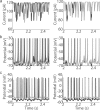
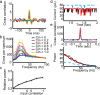
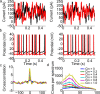
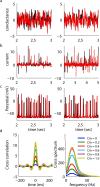
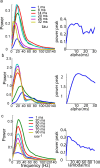
Oscillations are a common feature of odor-evoked and spontaneous activity in the olfactory system in vivo and in vitro and are thought to play an important role in information processing and memory in a variety of brain areas. Theoretical and experimental studies have described several mechanisms by which oscillations can be generated and synchronized. Here, we investigate the hypothesis that correlated noisy inputs are able to generate synchronous oscillations in olfactory bulb mitral cells in vitro. We consider several alternative mechanisms and conclude that olfactory bulb synchronous oscillations are likely to arise because of the response of uncoupled oscillating neurons to aperiodic but correlated inputs. This mechanism has been described theoretically, but we provide the first experimental evidence that such a mechanism may underlie synchronization in real neurons. In physiological experiments, we show that this mechanism can generate gamma-band oscillations in populations of olfactory bulb mitral cells. This mechanism synchronizes oscillatory firing by using shared fast fluctuations in stochastic inputs across neurons, without requiring any synaptic or electrical coupling. We discuss the properties and limitations of synchronization by this mechanism and suggest that it may underlie fast oscillations in many brain areas.
Figures


Similar articles
-
Interplay between local GABAergic interneurons and relay neurons generates gamma oscillations in the rat olfactory bulb.J Neurosci. 2004 May 5;24(18):4382-92. doi: 10.1523/JNEUROSCI.5570-03.2004. J Neurosci. 2004. PMID: 15128852 Free PMC article.
-
Synchronous Infra-Slow Oscillations Organize Ensembles of Accessory Olfactory Bulb Projection Neurons into Distinct Microcircuits.J Neurosci. 2020 May 20;40(21):4203-4218. doi: 10.1523/JNEUROSCI.2925-19.2020. Epub 2020 Apr 20. J Neurosci. 2020. PMID: 32312886 Free PMC article.
-
Circuit properties generating gamma oscillations in a network model of the olfactory bulb.J Neurophysiol. 2006 Apr;95(4):2678-91. doi: 10.1152/jn.01141.2005. Epub 2005 Dec 28. J Neurophysiol. 2006. PMID: 16381804
-
Adjusting neurophysiological computations in the adult olfactory bulb.Semin Cell Dev Biol. 2006 Aug;17(4):443-53. doi: 10.1016/j.semcdb.2006.04.011. Epub 2006 May 5. Semin Cell Dev Biol. 2006. PMID: 16757194 Review.
-
Electrical signaling in the olfactory bulb.Curr Opin Neurobiol. 2003 Aug;13(4):476-81. doi: 10.1016/s0959-4388(03)00092-8. Curr Opin Neurobiol. 2003. PMID: 12965296 Review.
Cited by
-
Top-Down Control of Inhibitory Granule Cells in the Main Olfactory Bulb Reshapes Neural Dynamics Giving Rise to a Diversity of Computations.Front Comput Neurosci. 2020 Jul 13;14:59. doi: 10.3389/fncom.2020.00059. eCollection 2020. Front Comput Neurosci. 2020. PMID: 32765248 Free PMC article.
-
Task Learning Promotes Plasticity of Interneuron Connectivity Maps in the Olfactory Bulb.J Neurosci. 2016 Aug 24;36(34):8856-71. doi: 10.1523/JNEUROSCI.0794-16.2016. J Neurosci. 2016. PMID: 27559168 Free PMC article.
-
Age-dependent adrenergic actions in the main olfactory bulb that could underlie an olfactory-sensitive period.J Neurophysiol. 2012 Oct;108(7):1999-2007. doi: 10.1152/jn.00322.2012. Epub 2012 Jul 18. J Neurophysiol. 2012. PMID: 22815401 Free PMC article.
-
Correlations in background activity control persistent state stability and allow execution of working memory tasks.Front Comput Neurosci. 2013 Oct 21;7:139. doi: 10.3389/fncom.2013.00139. eCollection 2013. Front Comput Neurosci. 2013. PMID: 24155714 Free PMC article.
-
What do we gain from gamma? Local dynamic gain modulation drives enhanced efficacy and efficiency of signal transmission.Front Hum Neurosci. 2010 Oct 21;4:185. doi: 10.3389/fnhum.2010.00185. eCollection 2010. Front Hum Neurosci. 2010. PMID: 21151350 Free PMC article.
References
-
- Abraham NM, Spors H, Carleton A, Margrie TW, Kuner T, Schaefer AT (2004). Maintaining accuracy at the expense of speed: stimulus similarity defines odor discrimination time in mice. Neuron 44:865–876. - PubMed
-
- Balu R, Larimer P, Strowbridge BW (2004). Phasic stimuli evoke precisely timed spikes in intermittently discharging mitral cells. J Neurophysiol 92:743–753. - PubMed
-
- Bressler SL, Freeman WJ (1980). Frequency analysis of olfactory system EEG in cat, rabbit, and rat. Electroencephalogr Clin Neurophysiol 50:19–24. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
