Alpha5GABAA receptors mediate the amnestic but not sedative-hypnotic effects of the general anesthetic etomidate
- PMID: 16597725
- PMCID: PMC6674127
- DOI: 10.1523/JNEUROSCI.5024-05.2006
Alpha5GABAA receptors mediate the amnestic but not sedative-hypnotic effects of the general anesthetic etomidate
Abstract
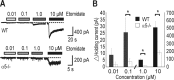
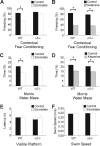
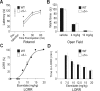
A fundamental objective of anesthesia research is to identify the receptors and brain regions that mediate the various behavioral components of the anesthetic state, including amnesia, immobility, and unconsciousness. Using complementary in vivo and in vitro approaches, we found that GABAA receptors that contain the alpha5 subunit (alpha5GABAARs) play a critical role in amnesia caused by the prototypic intravenous anesthetic etomidate. Whole-cell recordings from hippocampal pyramidal neurons showed that etomidate markedly increased a tonic inhibitory conductance generated by alpha5GABAARs, whereas synaptic transmission was only slightly enhanced. Long-term potentiation (LTP) of field EPSPs recorded in CA1 stratum radiatum was reduced by etomidate in wild-type (WT) but not alpha5 null mutant (alpha5-/-) mice. In addition, etomidate impaired memory performance of WT but not alpha5-/- mice for spatial and nonspatial hippocampal-dependent learning tasks. The brain concentration of etomidate associated with memory impairment in vivo was comparable with that which increased the tonic inhibitory conductance and blocked LTP in vitro. The alpha5-/- mice did not exhibit a generalized resistance to etomidate, in that the sedative-hypnotic effects measured with the rotarod, loss of righting reflex, and spontaneous motor activity were similar in WT and alpha5-/- mice. Deletion of the alpha5 subunit of the GABAARs reduced the amnestic but not the sedative-hypnotic properties of etomidate. Thus, the amnestic and sedative-hypnotic properties of etomidate can be dissociated on the basis of GABAAR subtype pharmacology.
Figures
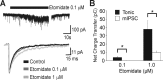

References
-
- Bai D, Zhu G, Pennefather P, Jackson MF, MacDonald JF, Orser BA (2001). Distinct functional and pharmacological properties of tonic and quantal inhibitory postsynaptic currents mediated by γ-aminobutyric acidA receptors in hippocampal neurons. Mol Pharmacol 59:814–824. - PubMed
-
- Barria A, Derkach V, Soderling T (1997). Identification of the Ca2+/calmodulin-dependentprotein kinase II regulatory phosphorylation site in the α-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate-type glutamate receptor. J Biol Chem 272:32727–32730. - PubMed
-
- Bliss TV, Collingridge GL (1993). A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31–39. - PubMed
-
- Campagna JA, Miller KW, Forman SA (2003). Mechanisms of actions of inhaled anesthetics. N Engl J Med 348:2110–2124. - PubMed
-
- Caraiscos VB, Elliott EM, You-Ten KE, Cheng VY, Belelli D, Newell JG, Jackson MF, Lambert JJ, Rosahl TW, Wafford KA, MacDonald JF, Orser BA (2004a). Tonic inhibition in mouse hippocampal CA1 pyramidal neurons is mediated by α5 subunit-containing γ-aminobutyric acid type A receptors. Proc Natl Acad Sci USA 101:3662–3667. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
