Calorimetric and structural studies of the nitric oxide carrier S-nitrosoglutathione bound to human glutathione transferase P1-1
- PMID: 16597834
- PMCID: PMC2242504
- DOI: 10.1110/ps.052055206
Calorimetric and structural studies of the nitric oxide carrier S-nitrosoglutathione bound to human glutathione transferase P1-1
Abstract
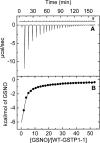
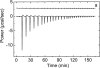
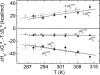
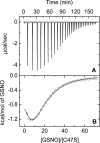
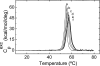
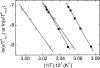
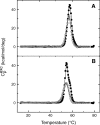
The nitric oxide molecule (NO) is involved in many important physiological processes and seems to be stabilized by reduced thiol species, such as S-nitrosoglutathione (GSNO). GSNO binds strongly to glutathione transferases, a major superfamily of detoxifying enzymes. We have determined the crystal structure of GSNO bound to dimeric human glutathione transferase P1-1 (hGSTP1-1) at 1.4 A resolution. The GSNO ligand binds in the active site with the nitrosyl moiety involved in multiple interactions with the protein. Isothermal titration calorimetry and differential scanning calorimetry (DSC) have been used to characterize the interaction of GSNO with the enzyme. The binding of GSNO to wild-type hGSTP1-1 induces a negative cooperativity with a kinetic process concomitant to the binding process occurring at more physiological temperatures. GSNO inhibits wild-type enzyme competitively at lower temperatures but covalently at higher temperatures, presumably by S-nitrosylation of a sulfhydryl group. The C47S mutation removes the covalent modification potential of the enzyme by GSNO. These results are consistent with a model in which the flexible helix alpha2 of hGST P1-1 must move sufficiently to allow chemical modification of Cys47. In contrast to wild-type enzyme, the C47S mutation induces a positive cooperativity toward GSNO binding. The DSC results show that the thermal stability of the mutant is slightly higher than wild type, consistent with helix alpha2 forming new interactions with the other subunit. All these results suggest that Cys47 plays a key role in intersubunit cooperativity and that under certain pathological conditions S-nitrosylation of Cys47 by GSNO is a likely physiological scenario.
Figures


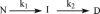
Similar articles
-
S-nitrosation of glutathione transferase p1-1 is controlled by the conformation of a dynamic active site helix.J Biol Chem. 2013 May 24;288(21):14973-84. doi: 10.1074/jbc.M113.462671. Epub 2013 Apr 9. J Biol Chem. 2013. PMID: 23572520 Free PMC article.
-
Human glutathione transferase P1-1 and nitric oxide carriers; a new role for an old enzyme.J Biol Chem. 2001 Nov 9;276(45):42138-45. doi: 10.1074/jbc.M102344200. Epub 2001 Aug 31. J Biol Chem. 2001. PMID: 11533048
-
Diuretic drug binding to human glutathione transferase P1-1: potential role of Cys-101 revealed in the double mutant C47S/Y108V.J Mol Recognit. 2011 Mar-Apr;24(2):220-34. doi: 10.1002/jmr.1040. J Mol Recognit. 2011. PMID: 20540076
-
S-nitrosoglutathione.Biochim Biophys Acta. 2013 May;1830(5):3173-81. doi: 10.1016/j.bbagen.2013.02.004. Epub 2013 Feb 14. Biochim Biophys Acta. 2013. PMID: 23416062 Free PMC article. Review.
-
Nitric oxide buffering and conditional nitric oxide release in stress response.J Exp Bot. 2018 Jun 19;69(14):3425-3438. doi: 10.1093/jxb/ery072. J Exp Bot. 2018. PMID: 29506191 Review.
Cited by
-
The good Samaritan glutathione-S-transferase P1: An evolving relationship in nitric oxide metabolism mediated by the direct interactions between multiple effector molecules.Redox Biol. 2023 Feb;59:102568. doi: 10.1016/j.redox.2022.102568. Epub 2022 Dec 15. Redox Biol. 2023. PMID: 36563536 Free PMC article. Review.
-
Evaluation of antioxidant, anti-inflammatory, anticancer activities and molecular docking of Moringa oleifera seed oil extract against experimental model of Ehrlich ascites carcinoma in Swiss female albino mice.BMC Complement Med Ther. 2023 Dec 14;23(1):457. doi: 10.1186/s12906-023-04279-z. BMC Complement Med Ther. 2023. PMID: 38098043 Free PMC article.
-
S-nitrosation of glutathione transferase p1-1 is controlled by the conformation of a dynamic active site helix.J Biol Chem. 2013 May 24;288(21):14973-84. doi: 10.1074/jbc.M113.462671. Epub 2013 Apr 9. J Biol Chem. 2013. PMID: 23572520 Free PMC article.
-
Influence of the H-site residue 108 on human glutathione transferase P1-1 ligand binding: structure-thermodynamic relationships and thermal stability.Protein Sci. 2009 Dec;18(12):2454-70. doi: 10.1002/pro.253. Protein Sci. 2009. PMID: 19780048 Free PMC article.
-
Redox compartmentalization in eukaryotic cells.Biochim Biophys Acta. 2008 Nov;1780(11):1273-90. doi: 10.1016/j.bbagen.2008.01.011. Epub 2008 Jan 26. Biochim Biophys Acta. 2008. PMID: 18267127 Free PMC article. Review.
References
-
- Battistoni A., Mazzetti A.P., Petruzzelli R., Muramatsu M., Federici G., Ricci G., Lo Bello M. 1995. Cytoplasmic and periplasmic production of human placental glutathione transferase in Escherichia coli Protein Expr. Purif. 6 579–587. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

