Transmissibility of cholera: in vivo-formed biofilms and their relationship to infectivity and persistence in the environment
- PMID: 16601099
- PMCID: PMC1458881
- DOI: 10.1073/pnas.0601277103
Transmissibility of cholera: in vivo-formed biofilms and their relationship to infectivity and persistence in the environment
Abstract
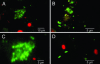
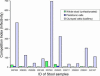
The factors that enhance the waterborne spread of bacterial epidemics and sustain the epidemic strain in nature are unclear. Although the epidemic diarrheal disease cholera is known to be transmitted by water contaminated with pathogenic Vibrio cholerae, routine isolation of pathogenic strains from aquatic environments is challenging. Here, we show that conditionally viable environmental cells (CVEC) of pathogenic V. cholerae that resist cultivation by conventional techniques exist in surface water as aggregates (biofilms) of partially dormant cells. Such CVEC can be recovered as fully virulent bacteria by inoculating the water into rabbit intestines. Furthermore, when V. cholerae shed in stools of cholera patients are inoculated in environmental water samples in the laboratory, the cells exhibit characteristics similar to CVEC, suggesting that CVEC are the infectious form of V. cholerae in water and that CVEC in nature may have been derived from human cholera stools. We also observed that stools from cholera patients contain a heterogeneous mixture of biofilm-like aggregates and free-swimming planktonic cells of V. cholerae. Estimation of the relative infectivity of these different forms of V. cholerae cells suggested that the enhanced infectivity of V. cholerae shed in human stools is largely due to the presence of clumps of cells that disperse in vivo, providing a high dose of the pathogen. The results of this study support a model of cholera transmission in which in vivo-formed biofilms contribute to enhanced infectivity and environmental persistence of pathogenic V. cholerae.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures

References
-
- Colwell R. R., Spira W. M. In: Cholera. Barua D., Greenough W. B. III, editors. New York: Plenum; 1992. pp. 107–127.
-
- Colwell R. R., Huq A. In: Vibrio cholerae and Cholera: Molecular to Global Perspectives. Wachsmuth I. K., Blake P. A., Olsvik O., editors. Washington, DC: Am. Soc. Microbiol; 1994. pp. 117–133.
-
- Pascual M., Rodo X., Ellner S. P., Colwell R. R., Bouma M. J. Science. 2000;289:1766–1769. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

