The kinetics of nucleated polymerizations at high concentrations: amyloid fibril formation near and above the "supercritical concentration"
- PMID: 16603497
- PMCID: PMC1479066
- DOI: 10.1529/biophysj.105.073767
The kinetics of nucleated polymerizations at high concentrations: amyloid fibril formation near and above the "supercritical concentration"
Abstract
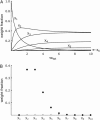
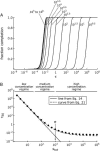
The formation of amyloid and other types of protein fibrils is thought to proceed by a nucleated polymerization mechanism. One of the most important features commonly associated with nucleated polymerizations is a strong dependence of the rate on the concentration. However, the dependence of fibril formation rates on concentration can weaken and nearly disappear as the concentration increases. Using numerical solutions to the rate equations for nucleated polymerization and analytical solutions to some limiting cases, we examine this phenomenon and show that it is caused by the concentration approaching and then exceeding the equilibrium constant for dissociation of monomers from species smaller than the nucleus, a quantity we have named the "supercritical concentration". When the concentration exceeds the supercritical concentration, the monomer, not the nucleus, is the highest-energy species on the fibril formation pathway, and the fibril formation reaction behaves initially like an irreversible polymerization. We also derive a relation that can be used in a straightforward method for determining the nucleus size and the supercritical concentration from experimental measurements of fibril formation rates.
Figures



References
-
- Sipe, J. D. 1994. Amyloidosis. Crit. Rev. Clin. Lab. Sci. 31:325–354. - PubMed
-
- Sipe, J. D., and A. S. Cohen. 2000. Review: history of the amyloid fibril. J. Struct. Biol. 130:88–98. - PubMed
-
- Selkoe, D. J. 2003. Folding proteins in fatal ways. Nature. 426:900–904. - PubMed
-
- Dobson, C. M. 2003. Protein folding and misfolding. Nature. 426:884–890. - PubMed
-
- Jarrett, J. T., and P. T. Lansbury. 1993. Seeding one-dimensional crystallization of amyloid: a pathogenic mechanism in Alzheimer's disease and scrapie. Cell. 73:1055–1058. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

