Bombesin inhibits alveolarization and promotes pulmonary fibrosis in newborn mice
- PMID: 16603607
- PMCID: PMC2662976
- DOI: 10.1164/rccm.200507-1014OC
Bombesin inhibits alveolarization and promotes pulmonary fibrosis in newborn mice
Erratum in
- Am J Respir Crit Care Med. 2006 Sep 1;174(5):616-7
Abstract
Rationale: Bombesin-like peptides promote fetal lung development. Normally, levels of mammalian bombesin (gastrin-releasing peptide [GRP]) drop postnatally, but these levels are elevated in newborns that develop bronchopulmonary dysplasia (BPD), a chronic lung disease characterized by arrested alveolarization. In premature baboons with BPD, antibombesin antibodies reduce lung injury and promote alveolarization.
Objectives: The present study tests whether exogenous bombesin or GRP given perinatally alters alveolar development in newborn mice.
Methods: Mice were given peptides intraperitoneally twice daily on Postnatal Days 1-3. On Day 14 lungs were inflation-fixed for histopathologic analyses of alveolarization.
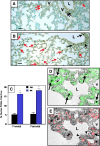
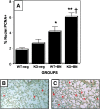
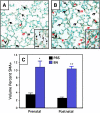
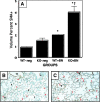
Measurements and main results: Bombesin had multiple effects on Day 14 lung, when alveolarization was about half complete. First, bombesin induced alveolar myofibroblast proliferation and increased alveolar wall thickness compared with saline-treated control animals. Second, bombesin diminished alveolarization in C57BL/6 (but not Swiss-Webster) mice. We used receptor-null mice to explore which receptors might mediate these effects. Compared with wild-type littermates, bombesin-treated GRP receptor (GRPR)-null mice had increased interstitial fibrosis but reduced defects in alveolarization. Neuromedin B (NMB) receptor-null and bombesin receptor subtype 3-null mice had the same responses as their wild-type littermates. GRP had the same effects as bombesin, whereas neither NMB nor a synthetic bombesin receptor type 3 ligand had any effect. All effects of GRP were abrogated in GRPR-null mice.
Conclusions: Bombesin/GRP can induce features of BPD, including interstitial fibrosis and diminished alveolarization. GRPR appears to mediate all effects of GRP, but only part of the bombesin effect on alveolarization, suggesting that novel receptors may mediate some effects of bombesin in newborn lung.
Figures
Similar articles
-
Bombesin-like peptides modulate alveolarization and angiogenesis in bronchopulmonary dysplasia.Am J Respir Crit Care Med. 2007 Nov 1;176(9):902-12. doi: 10.1164/rccm.200611-1734OC. Epub 2007 Jun 21. Am J Respir Crit Care Med. 2007. PMID: 17585105 Free PMC article.
-
The role of central gastrin-releasing peptide and neuromedin B receptors in the modulation of scratching behavior in rats.J Pharmacol Exp Ther. 2011 Jun;337(3):822-9. doi: 10.1124/jpet.111.178970. Epub 2011 Mar 18. J Pharmacol Exp Ther. 2011. PMID: 21421741 Free PMC article.
-
Evidence for autocrine actions of neuromedin B and gastrin-releasing peptide in non-small cell lung cancer.Pulm Pharmacol Ther. 1999;12(5):291-302. doi: 10.1006/pupt.1999.0210. Pulm Pharmacol Ther. 1999. PMID: 10545285
-
Two distinct receptor subtypes for mammalian bombesin-like peptides.Trends Neurosci. 1991 Dec;14(12):524-8. doi: 10.1016/0166-2236(91)90005-f. Trends Neurosci. 1991. PMID: 1726343 Review.
-
Molecular genetic analysis of two distinct receptors for mammalian bombesin-like peptides.J Natl Cancer Inst Monogr. 1992;(13):141-4. J Natl Cancer Inst Monogr. 1992. PMID: 1327030 Review.
Cited by
-
Maternal IL-1beta production prevents lung injury in a mouse model of bronchopulmonary dysplasia.Am J Respir Cell Mol Biol. 2010 Feb;42(2):149-60. doi: 10.1165/rcmb.2008-0287OC. Epub 2009 May 1. Am J Respir Cell Mol Biol. 2010. PMID: 19411613 Free PMC article.
-
IL-1beta disrupts postnatal lung morphogenesis in the mouse.Am J Respir Cell Mol Biol. 2007 Jan;36(1):32-42. doi: 10.1165/rcmb.2006-0116OC. Epub 2006 Aug 3. Am J Respir Cell Mol Biol. 2007. PMID: 16888287 Free PMC article.
-
Urine gastrin-releasing peptide in the first week correlates with bronchopulmonary dysplasia and post-prematurity respiratory disease.Pediatr Pulmonol. 2020 Apr;55(4):899-908. doi: 10.1002/ppul.24665. Epub 2020 Jan 29. Pediatr Pulmonol. 2020. PMID: 31995668 Free PMC article.
-
Bombesin-related peptides and their receptors: recent advances in their role in physiology and disease states.Curr Opin Endocrinol Diabetes Obes. 2008 Feb;15(1):58-64. doi: 10.1097/MED.0b013e3282f3709b. Curr Opin Endocrinol Diabetes Obes. 2008. PMID: 18185064 Free PMC article. Review.
-
Conditional overexpression of connective tissue growth factor disrupts postnatal lung development.Am J Respir Cell Mol Biol. 2010 May;42(5):552-63. doi: 10.1165/rcmb.2009-0068OC. Epub 2009 Jun 18. Am J Respir Cell Mol Biol. 2010. PMID: 19541844 Free PMC article.
References
-
- Sunday ME, Wolfe HJ, Roos BA, Chin WW, Spindel ER. Gastrin-releasing peptide gene expression in developing, hyperplastic, and neoplastic human thyroidal C-cells. Endocrinology 1988;122:1551–1558. - PubMed
-
- Sunday ME. Neuropeptides and lung development. In: McDonald JA, editor. Lung growth and development, 1st ed. New York: Marcel Dekker; 1997. pp. 401–494.
-
- Erspamer V. Amphibian skin peptides in mammals: looking ahead. Trends Neurosci 1983;6:200–201.
-
- McDonald TJ, Jornvall H, Nilsson G, Vagne M, Ghatei M, Bloom SR, Mutt V. Characterization of a gastrin-releasing peptide from porcine non-antral gastric tissue. Biochem Biophys Res Commun 1979;90:227–233. - PubMed
-
- Wharton J, Polak JM, Bloom SR, Ghatei MA, Solcia E, Brown MR, Pearse AGE. Bombesin-like immunoreactivity in the lung. Nature 1978;273:769–770. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

