Cholesterol-rich membrane microdomains mediate cell cycle arrest induced by Actinobacillus actinomycetemcomitans cytolethal-distending toxin
- PMID: 16611231
- PMCID: PMC4732718
- DOI: 10.1111/j.1462-5822.2005.00669.x
Cholesterol-rich membrane microdomains mediate cell cycle arrest induced by Actinobacillus actinomycetemcomitans cytolethal-distending toxin
Abstract
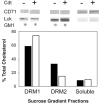
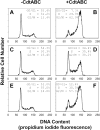
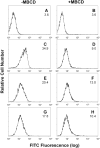
We have previously shown that Actinobacillus actinomycetemcomitans cytolethal-distending toxin (Cdt) is a potent immunosuppressive agent that induces G2/M arrest in human lymphocytes. In this study, we explored the possibility that Cdt-mediated immunotoxicity involves lipid membrane microdomains. We first determined that following treatment of Jurkat cells with Cdt holotoxin all three Cdt subunits localize to these microdomains. Laser confocal microscopy was employed to colocalize the subunits with GM1-enriched membrane regions which are characteristic of membrane rafts. Western blot analysis of isolated lipid rafts also demonstrated the presence of Cdt peptides. Cholesterol depletion, using methyl beta-cyclodextrin, protected cells from the ability of the Cdt holotoxin to induce G2 arrest. Moreover, cholesterol depletion reduced the ability of the toxin to associate with Jurkat cells. Thus, lipid raft integrity is vital to the action of Cdt on host cells. The implications of our observations with respect to Cdt mode of action are discussed.
Figures







References
-
- Allain CC, Poon LS, Chan CS, Richmond W, Fu PC. Enzymatic determination of total serum cholesterol. Clin Chem. 1974;20:470–475. - PubMed
-
- Alonso M, Millan J. The role of lipid rafts in signaling and membrane trafficking in T lymphocytes. J Cell Science. 2001;114:3957–3965. - PubMed
-
- Bartlett GR. Phosphorous assay in column chromatography. J Biol Chem. 1959;234:466–473. - PubMed
-
- Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Med Sci. 1959;8:911–917. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

