Members of the NIMA-related kinase family promote disassembly of cilia by multiple mechanisms
- PMID: 16611747
- PMCID: PMC1474788
- DOI: 10.1091/mbc.e05-05-0450
Members of the NIMA-related kinase family promote disassembly of cilia by multiple mechanisms
Abstract
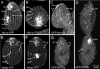
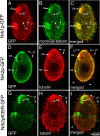
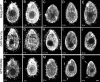
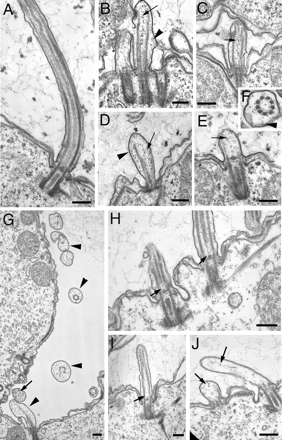
The genome of Tetrahymena thermophila contains 39 loci encoding NIMA-related kinases (NRKs), an extraordinarily large number for a unicellular organism. Evolutionary analyses grouped these sequences into several subfamilies, some of which have orthologues in animals, whereas others are protist specific. When overproduced, NRKs of three subfamilies caused rapid shortening of cilia. Ultrastructural studies revealed that each NRK triggered ciliary resorption by a distinct mechanism that involved preferential depolymerization of a subset of axonemal microtubules, at either the distal or proximal end. Overexpression of a kinase-inactive variant caused lengthening of cilia, indicating that constitutive NRK-mediated resorption regulates the length of cilia. Each NRK preferentially resorbed a distinct subset of cilia, depending on the location along the anteroposterior axis. We also show that normal Tetrahymena cells maintain unequal length cilia. We propose that ciliates used a large number of NRK paralogues to differentially regulate the length of specific subsets of cilia in the same cell.
Figures




References
-
- Berman S. A., Wilson N. F., Haas N. A., Lefebvre P. A. A novel MAP kinase regulates flagellar length in Chlamydomonas. Curr. Biol. 2003;13:1145–1149. - PubMed
-
- Bradley B. A., Quarmby L. M. A NIMA-related kinase, Cnk2p, regulates both flagellar length and cell size in Chlamydomonas. J. Cell Sci. 2005;118:3317–3326. - PubMed
-
- Bradley B. A., Wagner J. J., Quarmby L. M. Identification and sequence analysis of six new members of the NIMA-related kinase family in Chlamydomonas. J. Eukaryot. Microbiol. 2004;51:66–72. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

