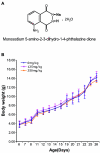
Retrovirus-induced oxidative stress with neuroimmunodegeneration is suppressed by antioxidant treatment with a refined monosodium alpha-luminol (Galavit)
- PMID: 16611916
- PMCID: PMC1472001
- DOI: 10.1128/JVI.80.9.4557-4569.2006
Retrovirus-induced oxidative stress with neuroimmunodegeneration is suppressed by antioxidant treatment with a refined monosodium alpha-luminol (Galavit)
Abstract
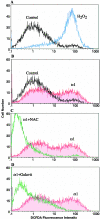
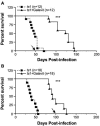
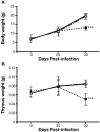
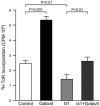
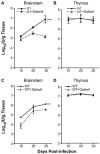
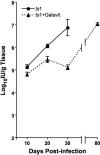
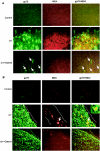
Oxidative stress is involved in many human neuroimmunodegenerative diseases, including human immunodeficiency virus disease/AIDS. The retrovirus ts1, a mutant of Moloney murine leukemia virus, causes oxidative stress and progressive neuro- and immunopathology in mice infected soon after birth. These pathological changes include spongiform neurodegeneration, astrogliosis, thymic atrophy, and T-cell depletion. Astrocytes and thymocytes are directly infected and killed by ts1. Neurons are not infected, but they also die, most likely as an indirect result of local glial infection. Cytopathic effects of ts1 infection in cultured astrocytes are associated with accumulation of the viral envelope precursor protein gPr80env in the endoplasmic reticulum (ER), which triggers ER stress and oxidative stress. We have reported (i) that activation of the Nrf2 transcription factor and upregulation of antioxidative defenses occurs in astrocytes infected with ts1 in vitro and (ii) that some ts1-infected astrocytes survive infection by mobilization of these pathways. Here, we show that treatment with a refined monosodium alpha-luminol (Galavit; GVT) suppresses oxidative stress and Nrf2 activation in cultured ts1-infected astrocytes. GVT treatment also inhibits the development of spongiform encephalopathy and gliosis in the central nervous system (CNS) in ts1-infected mice, preserves normal cytoarchitecture in the thymus, and delays paralysis, thymic atrophy, wasting, and death. GVT treatment of infected mice reduces ts1-induced oxidative stress, cell death, and pathogenesis in both the CNS and thymus of treated animals. These studies suggest that oxidative stress mediates ts1-induced neurodegeneration and T-cell loss.
Figures
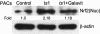



Similar articles
-
Neuroprotective effects of the drug GVT (monosodium luminol) are mediated by the stabilization of Nrf2 in astrocytes.Neurochem Int. 2010 May-Jun;56(6-7):780-8. doi: 10.1016/j.neuint.2010.02.017. Epub 2010 Mar 6. Neurochem Int. 2010. PMID: 20211212 Free PMC article.
-
The drug monosodium luminol (GVT) preserves thymic epithelial cell cytoarchitecture and allows thymocyte survival in mice infected with the T cell-tropic, cytopathic retrovirus ts1.Immunol Lett. 2009 Feb 21;122(2):159-69. doi: 10.1016/j.imlet.2008.12.009. Epub 2009 Feb 23. Immunol Lett. 2009. PMID: 19183564
-
The drug monosodium luminol (GVT) preserves crypt-villus epithelial organization and allows survival of intestinal T cells in mice infected with the ts1 retrovirus.Immunol Lett. 2009 Feb 21;122(2):150-8. doi: 10.1016/j.imlet.2008.12.012. Epub 2009 Jan 30. Immunol Lett. 2009. PMID: 19186189
-
Tsl and LP-BM5: a comparison of two murine retrovirus models for HIV.Viral Immunol. 2001;14(2):95-109. doi: 10.1089/088282401750234475. Viral Immunol. 2001. PMID: 11398815 Review.
-
Mouse Moloney leukemia virus infects microglia but not neurons even though it induces motor neuron disease.Mol Psychiatry. 1997 Mar;2(2):104-6. doi: 10.1038/sj.mp.4000219. Mol Psychiatry. 1997. PMID: 9106227 Review.
Cited by
-
PI3K/Akt/Nrf2 mediated cellular signaling and virus-host interactions: latest updates on the potential therapeutic management of SARS-CoV-2 infection.Front Mol Biosci. 2023 Jun 1;10:1158133. doi: 10.3389/fmolb.2023.1158133. eCollection 2023. Front Mol Biosci. 2023. PMID: 37325475 Free PMC article. Review.
-
The ubiquitin-proteasome system in spongiform degenerative disorders.Biochim Biophys Acta. 2008 Dec;1782(12):700-12. doi: 10.1016/j.bbadis.2008.08.006. Epub 2008 Aug 23. Biochim Biophys Acta. 2008. PMID: 18790052 Free PMC article. Review.
-
Attenuation of oxidative stress, inflammation and apoptosis by minocycline prevents retrovirus-induced neurodegeneration in mice.Brain Res. 2009 Aug 25;1286:174-84. doi: 10.1016/j.brainres.2009.06.007. Epub 2009 Jun 11. Brain Res. 2009. PMID: 19523933 Free PMC article.
-
Monosodium luminol reinstates redox homeostasis, improves cognition, mood and neurogenesis, and alleviates neuro- and systemic inflammation in a model of Gulf War Illness.Redox Biol. 2020 Jan;28:101389. doi: 10.1016/j.redox.2019.101389. Epub 2019 Nov 18. Redox Biol. 2020. PMID: 31778892 Free PMC article.
-
Pathophysiological basis and promise of experimental therapies for Gulf War Illness, a chronic neuropsychiatric syndrome in veterans.Psychopharmacology (Berl). 2023 Apr;240(4):673-697. doi: 10.1007/s00213-023-06319-5. Epub 2023 Feb 15. Psychopharmacology (Berl). 2023. PMID: 36790443 Review.
References
-
- Ball, J., J. A. McCarter, and S. M. Sunderland. 1973. Evidence for helper independent murine sarcoma virus. I. Segregation of replication-defective and transformation-defective viruses. Virology 56:268-284. - PubMed
-
- Bensaad, K., and K. H. Vousden. 2005. Savior and slayer: the two faces of p53. Nat. Med. 11:1278-1279. - PubMed
-
- Betteridge, D. J. 2000. What is oxidative stress? Metabolism 49:3-8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

