Identification of a structural motif crucial for infectivity of hepatitis B viruses
- PMID: 16618937
- PMCID: PMC1458949
- DOI: 10.1073/pnas.0509765103
Identification of a structural motif crucial for infectivity of hepatitis B viruses
Abstract
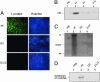
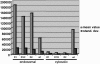
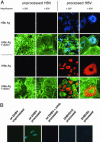
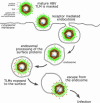
Infectious entry of hepatitis B viruses (HBV) has nonconventional facets. Here we analyzed whether a cell-permeable peptide [translocation motif (TLM)] identified within the surface protein of human HBV is a general feature of all hepadnaviruses and plays a role in the viral life cycle. Surface proteins of all hepadnaviruses contain conserved functional TLMs. Genetic inactivation of the duck HBV TLMs does not interfere with viral morphogenesis; however, these mutants are noninfectious. TLM mutant viruses bind to cells and are taken up into the endosomal compartment, but they cannot escape from endosomes. Processing of surface protein by endosomal proteases induces their exposure on the virus surface. This unmasking of TLMs mediates translocation of viral particles across the endosomal membrane into the cytosol, a prerequisite for productive infection. The ability of unmasked TLMs to translocate processed HBV particles across cellular membranes was shown by confocal immunofluorescence microscopy and by infection of nonpermissive cell lines with HBV processed in vitro with endosomal lysate. Based on these data, we propose an infectious entry mechanism unique for hepadnaviruses that involves virus internalization by receptor-mediated endocytosis followed by processing of surface protein in endosomes. This processing activates the function of TLMs that are essential for viral particle translocation through the endosomal membrane into the cytosol and productive infection.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

