Eye cancer: unique insights into oncogenesis: the Cogan Lecture
- PMID: 16638975
- PMCID: PMC1769553
- DOI: 10.1167/iovs.05-1291
Eye cancer: unique insights into oncogenesis: the Cogan Lecture
Figures




Similar articles
-
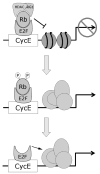
Expression patterns of cyclin D1 and related proteins regulating G1-S phase transition in uveal melanoma and retinoblastoma.Br J Ophthalmol. 1998 Aug;82(8):961-70. doi: 10.1136/bjo.82.8.961. Br J Ophthalmol. 1998. PMID: 9828785 Free PMC article.
-
Retinoblastoma in the 20th century: past success and future challenges the Weisenfeld lecture.Invest Ophthalmol Vis Sci. 2005 Aug;46(8):2683-91. doi: 10.1167/iovs.04-1462. Invest Ophthalmol Vis Sci. 2005. PMID: 16043839 No abstract available.
-
The molecular biology of retinoblastoma.Ocul Immunol Inflamm. 2001 Mar;9(1):1-8. doi: 10.1076/ocii.9.1.1.3984. Ocul Immunol Inflamm. 2001. PMID: 11262663 Review.
-
International Society for Eye Research--XIXth Biennial Meeting.IDrugs. 2010 Sep;13(9):619-21. IDrugs. 2010. PMID: 20799143
-
Update on Ophthalmic Oncology 2014: Retinoblastoma and Uveal Melanoma.Asia Pac J Ophthalmol (Phila). 2016 Sep-Oct;5(5):368-82. doi: 10.1097/APO.0000000000000213. Asia Pac J Ophthalmol (Phila). 2016. PMID: 27632029 Review.
Cited by
-
Enucleated globes with choroidal melanoma: A retrospective histopathological study and correlation with cytogenetic profile in 2 eye centers.Ann Med Surg (Lond). 2020 Jun 1;55:227-233. doi: 10.1016/j.amsu.2020.05.003. eCollection 2020 Jul. Ann Med Surg (Lond). 2020. PMID: 32518647 Free PMC article.
-
An alternative hypothesis for observed mortality rates due to metastasis after treatment of choroidal melanomas of different sizes.Trans Am Ophthalmol Soc. 2007;105:54-59; discussion 59-60. Trans Am Ophthalmol Soc. 2007. PMID: 18427594 Free PMC article. Review.
-
Cytogenetic results of choroidal nevus growth into melanoma in 55 consecutive cases.Saudi J Ophthalmol. 2018 Jan-Mar;32(1):28-32. doi: 10.1016/j.sjopt.2018.02.004. Epub 2018 Feb 15. Saudi J Ophthalmol. 2018. PMID: 29755268 Free PMC article.
-
Retinoblastoma. Fifty years of progress. The LXXI Edward Jackson Memorial Lecture.Am J Ophthalmol. 2014 Nov;158(5):875-91. doi: 10.1016/j.ajo.2014.07.025. Epub 2014 Jul 24. Am J Ophthalmol. 2014. PMID: 25065496 Free PMC article.
-
Uveal melanoma: molecular pattern, clinical features, and radiation response.Am J Ophthalmol. 2012 Aug;154(2):227-232.e2. doi: 10.1016/j.ajo.2012.02.022. Epub 2012 Apr 27. Am J Ophthalmol. 2012. PMID: 22541662 Free PMC article.
References
-
- Harbour JW. Retinoblastoma: pathogenesis and diagnosis. In: Char DH, editor. Tumors of the Eye and Orbit. Philadelphia: BC Decker; 2001. pp. 253–265.
-
- Neel JV, Falls HF. The rate of mutation of the gene responsible for retinoblastoma in man. Science. 1951;114:419–422. - PubMed
-
- Friend SH, Bernards R, Rogelj S, et al. A human DNA segment with properties of the gene that predisposes to retinoblastoma and osteosarcoma. Nature. 1986;323:643–646. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

