Astrocytes derived from glial-restricted precursors promote spinal cord repair
- PMID: 16643674
- PMCID: PMC1561531
- DOI: 10.1186/jbiol35
Astrocytes derived from glial-restricted precursors promote spinal cord repair
Abstract
Background: Transplantation of embryonic stem or neural progenitor cells is an attractive strategy for repair of the injured central nervous system. Transplantation of these cells alone to acute spinal cord injuries has not, however, resulted in robust axon regeneration beyond the sites of injury. This may be due to progenitors differentiating to cell types that support axon growth poorly and/or their inability to modify the inhibitory environment of adult central nervous system (CNS) injuries. We reasoned therefore that pre-differentiation of embryonic neural precursors to astrocytes, which are thought to support axon growth in the injured immature CNS, would be more beneficial for CNS repair.
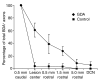
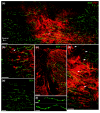
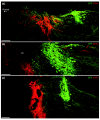
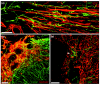
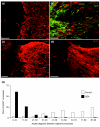
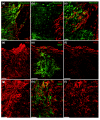
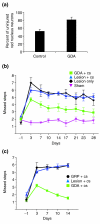
Results: Transplantation of astrocytes derived from embryonic glial-restricted precursors (GRPs) promoted robust axon growth and restoration of locomotor function after acute transection injuries of the adult rat spinal cord. Transplantation of GRP-derived astrocytes (GDAs) into dorsal column injuries promoted growth of over 60% of ascending dorsal column axons into the centers of the lesions, with 66% of these axons extending beyond the injury sites. Grid-walk analysis of GDA-transplanted rats with rubrospinal tract injuries revealed significant improvements in locomotor function. GDA transplantation also induced a striking realignment of injured tissue, suppressed initial scarring and rescued axotomized CNS neurons with cut axons from atrophy. In sharp contrast, undifferentiated GRPs failed to suppress scar formation or support axon growth and locomotor recovery.
Conclusion: Pre-differentiation of glial precursors into GDAs before transplantation into spinal cord injuries leads to significantly improved outcomes over precursor cell transplantation, providing both a novel strategy and a highly effective new cell type for repairing CNS injuries.
Figures



Comment in
-
Building bridges with astrocytes for spinal cord repair.J Biol. 2006;5(3):6. doi: 10.1186/jbiol40. Epub 2006 May 9. J Biol. 2006. PMID: 16684372 Free PMC article.
References
-
- Moreau-Fauvarque C, Kumanogoh A, Camand E, Jaillard C, Barbin G, Boquet I, Love C, Jones EY, Kikutani H, Lubetzki C, et al. The transmembrane semaphorin Sema4D/CD100, an inhibitor of axonal growth, is expressed on oligodendrocytes and upregulated after CNS lesion. J Neurosci. 2003;23:9229–9239. - PMC - PubMed
-
- Tang X, Davies JE, Davies SJ. Changes in distribution, cell associations, and protein expression levels of NG2, neurocan, phosphacan, brevican, versican V2, and tenascin-C during acute to chronic maturation of spinal cord scar tissue. J Neurosci Res. 2003;71:427–444. doi: 10.1002/jnr.10523. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

