Protective immune responses against West Nile virus are primed by distinct complement activation pathways
- PMID: 16651386
- PMCID: PMC2121216
- DOI: 10.1084/jem.20052388
Protective immune responses against West Nile virus are primed by distinct complement activation pathways
Abstract
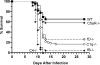
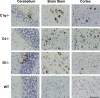
West Nile virus (WNV) causes a severe infection of the central nervous system in several vertebrate animals including humans. Prior studies have shown that complement plays a critical role in controlling WNV infection in complement (C) 3(-/-) and complement receptor 1/2(-/-) mice. Here, we dissect the contributions of the individual complement activation pathways to the protection from WNV disease. Genetic deficiencies in C1q, C4, factor B, or factor D all resulted in increased mortality in mice, suggesting that all activation pathways function together to limit WNV spread. In the absence of alternative pathway complement activation, WNV disseminated into the central nervous system at earlier times and was associated with reduced CD8+ T cell responses yet near normal anti-WNV antibody profiles. Animals lacking the classical and lectin pathways had deficits in both B and T cell responses to WNV. Finally, and somewhat surprisingly, C1q was required for productive infection in the spleen but not for development of adaptive immune responses after WNV infection. Our results suggest that individual pathways of complement activation control WNV infection by priming adaptive immune responses through distinct mechanisms.
Figures

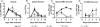
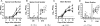
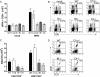
References
-
- Carroll, M.C. 2004. The complement system in regulation of adaptive immunity. Nat. Immunol. 5:981–986. - PubMed
-
- Kopf, M., B. Abel, A. Gallimore, M. Carroll, and M.F. Bachmann. 2002. Complement component C3 promotes T-cell priming and lung migration to control acute influenza virus infection. Nat. Med. 8:373–378. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

