Analysis of FimX, a phosphodiesterase that governs twitching motility in Pseudomonas aeruginosa
- PMID: 16677312
- PMCID: PMC3609419
- DOI: 10.1111/j.1365-2958.2006.05156.x
Analysis of FimX, a phosphodiesterase that governs twitching motility in Pseudomonas aeruginosa
Abstract
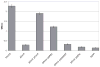
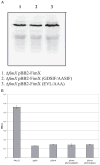
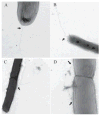
Type IV pili (Tfp) are polar surface structures of Pseudomonas aeruginosa required for twitching motility, biofilm formation and adherence. One protein required for the assembly of tfp is FimX, which possesses both GGDEF and EAL domains characteristic of diguanylate cyclases and phosphodiesterases respectively. In this work we demonstrate that FimX has phosphodiesterase activity towards bis-(3'-5')-cyclic dimeric guanosine monophosphate (c-di-GMP), but does not show diguanylate cyclase activity. Instead, the imperfect GGDEF domain of FimX likely serves to activate phosphodiesterase activity when bound to GTP, as has recently been described for the Caulobacter crescentus composite GGDEF-EAL protein, CC3396. Bacteria expressing FimX in which either the GGDEF or EAL domain is deleted or mutated have phenotypes indistinguishable from a DeltafimX strain, demonstrating the importance of both domains to function. Previous work has shown that FimX localizes to the bacterial pole. In this work we show that restriction of FimX to a single pole requires intact GGDEF and EAL domains. Deletion of the amino-terminal REC domain of FimX, which contains a putative polar localization signal, results in a protein that still supports intermediate levels of pilus assembly and function. RFP-FimXDeltaREC, unlike RFP-FimX, is no longer localized to the bacterial pole, while transmission electron microscopy shows that surface pili can originate from non-polar sites in this mutant. Although DeltafimX mutants show limited in vitro cytotoxicity, they are as virulent as the wild-type strain in a murine model of acute pneumonia.
Figures





References
-
- Aldridge P, Jenal U. Cell cycle-dependent degradation of a flagellar motor component requires a novel-type response regulator. Mol Microbiol. 1999;32:379–391. - PubMed
-
- Ausmees N, Jonsson H, Hoglund S, Ljunggren HG, Lindberg M. Structural and putative regulatory genes involved in cellulose synthesis in Rhizobium leguminosarum bv. trifolii. Microbiology. 1999;145:1253–1262. - PubMed
-
- Bobrov AG, Kirillina O, Perry RD. The phosphodiesterase activity of the HmsP EAL domain is required for negative regulation of biofilm formation in Yersinia pestis. FEMS Microbiol Lett. 2005;247:123–130. - PubMed
-
- Bradley DE. A function of Pseudomonas aeruginosa PAO pili: twitching motility. Can J Microbiol. 1980;26:146–154. - PubMed
-
- Brookman JL, Stott AJ, Cheeseman PJ, Burns NR, Adams SE, Kingsman AJ, Gull K. An immunological analysis of Ty1 virus-like particle structure. Virology. 1995;207:59–67. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

