Functional significance of differential eNOS translocation
- PMID: 16679407
- PMCID: PMC1629085
- DOI: 10.1152/ajpheart.00370.2006
Functional significance of differential eNOS translocation
Abstract
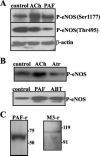
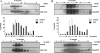
Nitric oxide (NO) regulates flow and permeability. ACh and platelet-activating factor (PAF) lead to endothelial NO synthase (eNOS) phosphorylation and NO release. While ACh causes only vasodilation, PAF induces vasoconstriction and hyperpermeability. The key differential signaling mechanisms for discriminating between vasodilation and hyperpermeability are unknown. We tested the hypothesis that differential translocation may serve as a regulatory mechanism of eNOS to determine specific vascular responses. We used ECV-304 cells permanently transfected with eNOS-green fluorescent protein (ECVeNOS-GFP) and demonstrated that the agonists activate eNOS and reproduce their characteristic endothelial permeability effects in these cells. We evaluated eNOS localization by lipid raft analysis and immunofluorescence microscopy. After PAF and ACh, eNOS moves away from caveolae. eNOS distributes both in the plasma membrane and Golgi in control cells. ACh (10(-5) M, 10(-4) M) translocated eNOS preferentially to the trans-Golgi network (TGN) and PAF (10(-7) M) preferentially to the cytosol. We suggest that PAF-induced eNOS translocation preferentially to cytosol reflects a differential signaling mechanism related to changes in permeability, whereas ACh-induced eNOS translocation to the TGN is related to vasodilation.
Figures





Comment in
-
New insights into eNOS signaling in microvascular permeability.Am J Physiol Heart Circ Physiol. 2006 Sep;291(3):H1029-31. doi: 10.1152/ajpheart.00509.2006. Epub 2006 May 26. Am J Physiol Heart Circ Physiol. 2006. PMID: 16731639 No abstract available.
References
-
- Breslin JW, Pappas PJ, Cerveira JJ, Hobson RW, II, Durán WN. VEGF increases endothelial permeability by separate signaling pathways involving ERK-1/2 and nitric oxide. Am J Physiol Heart Circ Physiol. 2003;284:H92–H100. - PubMed
-
- Corson MA, James NL, Latta SE, Nerem RM, Berk BC, Harrison DG. Phosphorylation of endothelial nitric oxide synthase in response to fluid shear stress. Circ Res. 1996;79:984–991. - PubMed
-
- Dillon PK, Durán WN. Effect of platelet-activating factor on micro-vascular permselectivity: dose-response relations and pathways of action in the hamster cheek pouch microcirculation. Circ Res. 1988;62:732–740. - PubMed
-
- Durán WN, Dillon PK. Acute microcirculatory effects of platelet-activating factor. J Lipid Mediat. 1990;2(Suppl):S215–S227. - PubMed
-
- Durán WN, Seyama A, Yoshimura K, González DR, Jara PI, Figueroa XF, Boric MP. Stimulation of NO production and of eNOS phosphorylation in the microcirculation in vivo. Microvasc Res. 2000;60:104–111. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

