Use of an integrated clinical trial database to evaluate the effect of timing of drotrecogin alfa (activated) treatment in severe sepsis
- PMID: 16684364
- PMCID: PMC1550949
- DOI: 10.1186/cc4909
Use of an integrated clinical trial database to evaluate the effect of timing of drotrecogin alfa (activated) treatment in severe sepsis
Abstract
Introduction: Several studies have indicated that early identification and treatment of patients with severe sepsis using standard supportive care improves outcomes. Earlier treatment with drotrecogin alfa (activated) (DrotAA) may also improve outcomes in severe sepsis. Using a recently constructed integrated severe sepsis database, our objectives in this study were to describe the influence of baseline clinical characteristics on timing of DrotAA treatment in patients with severe sepsis, to evaluate the efficacy of DrotAA with respect to timing of administration, and to examine the association between early intervention with DrotAA and patient outcomes, using adjustments for imbalances.
Methods: The database comprises data from 4,459 patients with severe sepsis (DrotAA, n = 3,228; placebo, n = 1,231) included in five clinical trials conducted in tertiary care institutions in 28 countries. Placebo data came only from randomized trials, whereas data for the DrotAA group came from randomized (PROWESS) and open-label/observational (ENHANCE) trials.
Results: Increased time-to-treatment with DrotAA was significantly associated with more organ dysfunction, greater need of mechanical ventilation, vasopressor use, or recent surgery. Earlier treatment was associated with higher baseline Acute Physiology and Chronic Health Evaluation (APACHE II) scores. Adjusted and unadjusted survival analyses suggested that compared with placebo, DrotAA treatment provided a potential survival benefit, regardless of time to treatment. Survival curves of DrotAA patients treated early compared with those treated late began to separate at 14 days. By 28 days, patients treated earlier had higher survival than those treated later (76.4% versus 73.5%, p = 0.03). Sepsis-induced multiorgan dysfunction was the most common cause of death followed by refractory shock and respiratory failure. Modeling of the treatment effect, as a function of time to treatment, suggested increased benefit with earlier treatment.
Conclusion: Using an integrated database of five severe sepsis trials and appropriate statistical adjustments to reduce sources of potential bias, earlier treatment with DrotAA seemed to be associated with a lower risk-adjusted mortality than later treatment. These data suggest that earlier treatment with DrotAA may provide most benefit for appropriate patients.
Figures

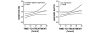
Comment in
-
Timing of drotrecogin alfa (activated) treatment in severe sepsis.Crit Care. 2006;10(4):419; author reply 419. doi: 10.1186/cc5010. Crit Care. 2006. PMID: 16934134 Free PMC article. No abstract available.
References
-
- Vincent JL, Bernard GR, Beale R, Doig C, Putensen C, Dhainaut JF, Artigas A, Fumagalli R, Macias W, Wright T, et al. Drotrecogin alfa (activated) treatment in severe sepsis from the global open-label trial ENHANCE. Crit Care Med. 2005;33:2266–2277. doi: 10.1097/01.CCM.0000181729.46010.83. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

