Does CO2 permeate through aquaporin-1?
- PMID: 16698771
- PMCID: PMC1563782
- DOI: 10.1529/biophysj.106.081406
Does CO2 permeate through aquaporin-1?
Abstract
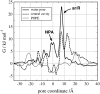
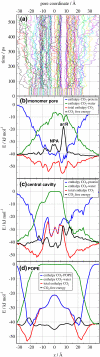
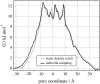
Aquaporins facilitate water permeation across biological membranes. Additionally, glycerol and other small neutral solutes are permeated by related aquaglyceroporins. The role of aquaporins in gas permeation has been a long-standing and controversially discussed issue. We present an extensive set of atomistic molecular dynamics simulations that address the question of CO(2) permeation through human aquaporin-1. Free energy profiles derived from the simulations display a barrier of approximately 23 kJ/mol in the aromatic/arginine constriction region of the water pore, whereas a barrier of approximately 4 kJ/mol was observed for a palmitoyloleoylphosphatidylethanolamine lipid bilayer membrane. The results indicate that significant aquaporin-1-mediated CO(2) permeation is to be expected only in membranes with a low intrinsic CO(2) permeability.
Figures

Similar articles
-
Mechanism of selectivity in aquaporins and aquaglyceroporins.Proc Natl Acad Sci U S A. 2008 Jan 29;105(4):1198-203. doi: 10.1073/pnas.0707662104. Epub 2008 Jan 17. Proc Natl Acad Sci U S A. 2008. PMID: 18202181 Free PMC article.
-
Exploring gas permeability of cellular membranes and membrane channels with molecular dynamics.J Struct Biol. 2007 Mar;157(3):534-44. doi: 10.1016/j.jsb.2006.11.008. Epub 2007 Jan 4. J Struct Biol. 2007. PMID: 17306562
-
Dynamics and energetics of permeation through aquaporins. What do we learn from molecular dynamics simulations?Handb Exp Pharmacol. 2009;(190):57-76. doi: 10.1007/978-3-540-79885-9_3. Handb Exp Pharmacol. 2009. PMID: 19096772 Review.
-
Molecular dynamics study of the archaeal aquaporin AqpM.BMC Genomics. 2011 Dec 22;12 Suppl 4(Suppl 4):S8. doi: 10.1186/1471-2164-12-S4-S8. Epub 2011 Dec 22. BMC Genomics. 2011. PMID: 22369250 Free PMC article.
-
The dynamics and energetics of water permeation and proton exclusion in aquaporins.Curr Opin Struct Biol. 2005 Apr;15(2):176-83. doi: 10.1016/j.sbi.2005.02.003. Curr Opin Struct Biol. 2005. PMID: 15837176 Review.
Cited by
-
Molecular dynamics of water in the neighborhood of aquaporins.Eur Biophys J. 2013 Apr;42(4):223-39. doi: 10.1007/s00249-012-0880-y. Epub 2012 Dec 29. Eur Biophys J. 2013. PMID: 23274929 Review.
-
Presence of Intra-helical Salt-Bridge in Loop E Half-Helix Can Influence the Transport Properties of AQP1 and GlpF Channels: Molecular Dynamics Simulations of In Silico Mutants.J Membr Biol. 2019 Feb;252(1):17-29. doi: 10.1007/s00232-018-0054-7. Epub 2018 Nov 23. J Membr Biol. 2019. PMID: 30470864
-
Extra- and intracellular unstirred layer effects in measurements of CO2 diffusion across membranes--a novel approach applied to the mass spectrometric 18O technique for red blood cells.J Physiol. 2009 Mar 15;587(Pt 6):1153-67. doi: 10.1113/jphysiol.2008.165027. Epub 2009 Jan 12. J Physiol. 2009. PMID: 19139045 Free PMC article.
-
Amino Acid Substitutions in Loop C of Arabidopsis PIP2 Aquaporins Alters the Permeability of CO2.Plant Cell Environ. 2025 Sep;48(9):6835-6846. doi: 10.1111/pce.15635. Epub 2025 Jun 3. Plant Cell Environ. 2025. PMID: 40459249 Free PMC article.
-
Exploring transmembrane diffusion pathways with molecular dynamics.Physiology (Bethesda). 2010 Jun;25(3):142-54. doi: 10.1152/physiol.00046.2009. Physiology (Bethesda). 2010. PMID: 20551228 Free PMC article. Review.
References
-
- Preston, G. M., T. P. Carroll, W. B. Guggino, and P. Agre. 1992. Appearance of water channels in Xenopus oocytes expressing red-cell CHIP28 protein. Science. 256:385–387. - PubMed
-
- Zeidel, M. L., S. V. Ambudkar, B. L. Smith, and P. Agre. 1992. Reconstitution of functional water channels in liposomes containing purified red-cell CHIP28 protein. Biochemistry. 31:7436–7440. - PubMed
-
- Maurel, C., J. Reizer, J. I. Schroeder, M. J. Chrispeels, and M. H. Saier. 1994. Functional characterization of the Eschericia coli glycerol facilitator, GlpF, in Xenopus oocytes. J. Biol. Chem. 269:11869–11872. - PubMed
-
- Agre, P., M. Bonhivers, and M. J. Borgnia. 1998. The aquaporins, blueprints for cellular plumbing systems. J. Biol. Chem. 273:14659–14662. - PubMed
-
- Deen, P. M. T., and C. H. van Os. 1998. Epithelial aquaporins. Curr. Opin. Cell Biol. 10:435–442. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

