Frequency-specific and D2 receptor-mediated inhibition of glutamate release by retrograde endocannabinoid signaling
- PMID: 16698932
- PMCID: PMC1472459
- DOI: 10.1073/pnas.0510797103
Frequency-specific and D2 receptor-mediated inhibition of glutamate release by retrograde endocannabinoid signaling
Abstract
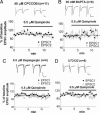
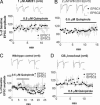
The mechanisms underlying modulation of corticostriatal synaptic transmission by D2-like receptors (D2Rs) have been controversial. A recent study suggested that D2Rs inhibit glutamate release at this synapse, but only during high-frequency synaptic activation. Because the release of postsynaptic endocannabinoids (eCBs), which act as retrograde messengers to inhibit presynaptic glutamate release, can be triggered by D2R activation and intense synaptic activation, such a mechanism could mediate dopaminergic modulation of corticostriatal transmission. Here, we show that D2R activation reduces excitatory transmission onto striatal medium spiny neurons at a stimulation frequency of 20 Hz but not at 1 Hz. This form of inhibition requires CB1 receptor activation, as evidenced by the fact that it is blocked by AM251 [N-(piperidin-1-yl)-1-(2,4-dichlorophenyl)-5-(4-chlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide], a CB1 antagonist, and is absent in CB1 knockout mice. It is also blocked by postsynaptic intracellular calcium chelation, by group I metabotropic glutamate receptor antagonism, and by inhibition of postsynaptic phospholipase C. These results demonstrate a previously unrecognized role for retrograde eCB signaling in reversible and frequency-specific inhibition of glutamate release by the activation of striatal D2Rs.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures
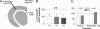


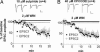
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

