Gamma-tubulin is essential for microtubule organization and development in Arabidopsis
- PMID: 16698945
- PMCID: PMC1475493
- DOI: 10.1105/tpc.105.039644
Gamma-tubulin is essential for microtubule organization and development in Arabidopsis
Abstract
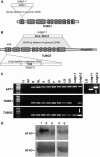
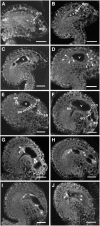
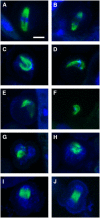
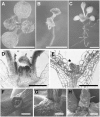
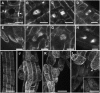
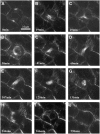
The process of microtubule nucleation in plant cells is still a major question in plant cell biology. gamma-Tubulin is known as one of the key molecular players for microtubule nucleation in animal and fungal cells. Here, we provide genetic evidence that in Arabidopsis thaliana, gamma-tubulin is required for the formation of spindle, phragmoplast, and cortical microtubule arrays. We used a reverse genetics approach to investigate the role of the two Arabidopsis gamma-tubulin genes in plant development and in the formation of microtubule arrays. Isolation of mutants in each gene and analysis of two combinations of gamma-tubulin double mutants showed that the two genes have redundant functions. The first combination is lethal at the gametophytic stage. Disruption of both gamma-tubulin genes causes aberrant spindle and phragmoplast structures and alters nuclear division in gametophytes. The second combination of gamma-tubulin alleles affects late seedling development, ultimately leading to lethality 3 weeks after germination. This partially viable mutant combination enabled us to follow dynamically the effects of gamma-tubulin depletion on microtubule arrays in dividing cells using a green fluorescent protein marker. These results establish the central role of gamma-tubulin in the formation and organization of microtubule arrays in Arabidopsis.
Figures


References
-
- Alexander, M.P. (1969). Differential staining of aborted and non aborted pollen. Stain Technol. 44 117–122. - PubMed
-
- Alonso, J.M., et al. (2003). Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301 653–657. - PubMed
-
- Arabidopsis Genome Initiative (2000). Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408 796–815. - PubMed
-
- Azimzadeh, J., Traas, J., and Pastuglia, M. (2001). Molecular aspects of microtubule dynamics in plants. Curr. Opin. Plant Biol. 4 513–519. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

