Sequence requirements for localization of human cytomegalovirus tegument protein pp28 to the virus assembly compartment and for assembly of infectious virus
- PMID: 16699042
- PMCID: PMC1472139
- DOI: 10.1128/JVI.02630-05
Sequence requirements for localization of human cytomegalovirus tegument protein pp28 to the virus assembly compartment and for assembly of infectious virus
Abstract
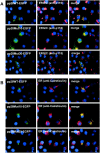
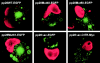
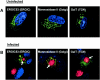
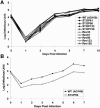
The human cytomegalovirus UL99 open reading frame encodes a 190-amino-acid (aa) tegument protein, pp28, that is myristoylated and phosphorylated. pp28 is essential for assembly of infectious virus, and nonenveloped virions accumulate in the cytoplasm of cells infected with recombinant viruses with a UL99 deletion. pp28 is localized to the endoplasmic reticulum-Golgi intermediate compartment (ERGIC) in transfected cells, while in infected cells, it is localized together with other virion proteins in a juxtanuclear compartment termed the assembly compartment (AC). We investigated the sequence requirements for pp28 trafficking to the AC and assembly of infectious virus. Our studies indicated that the first 30 to 35 aa were required for localization of pp28 to the ERGIC in transfected cells. Mutant forms of pp28 containing only the first 35 aa localized with other virion structural proteins to cytoplasmic compartments early in infection, but localization to the AC at late times required a minimum of 50 aa. In agreement with previous reports, we demonstrated that the deletion of a cluster of acidic amino acids (aa 44 to 59) prevented wild-type trafficking of pp28 and recovery of infectious virus. A recombinant virus expressing only the first 50 aa was replication competent, and this mutant, pp28, localized to the AC in cells infected with this virus. These findings argued that localization of pp28 to the AC was essential for assembly of infectious virus and raised the possibility that amino acids in the amino terminus of pp28 have additional roles in the envelopment and assembly of the virion other than simply localizing pp28 to the AC.
Figures





References
-
- Andreoni, M., M. Faircloth, L. Vugler, and W. J. Britt. 1989. A rapid microneutralization assay for the measurement of neutralizing antibody reactive with human cytomegalovirus. J. Virol. Methods 23:157-167. - PubMed
-
- Appenzeller, C., H. Andersson, F. Kappeler, and H. P. Hauri. 1999. The lectin ERGIC-53 is a cargo transport receptor for glycoproteins. Nat. Cell Biol. 1:330-334. - PubMed
-
- Bonifacino, J. S., and L. M. Traub. 2003. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 72:395-447. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

