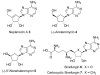
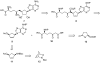
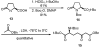
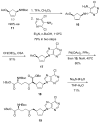
Substrate-dependent dihydroxylation of substituted cyclopentenes: toward the syntheses of carbocyclic sinefungin and noraristeromycin
- PMID: 16709056
- PMCID: PMC2652561
- DOI: 10.1021/jo060224l
Substrate-dependent dihydroxylation of substituted cyclopentenes: toward the syntheses of carbocyclic sinefungin and noraristeromycin
Abstract
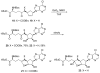
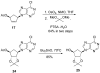
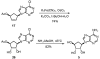
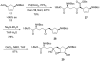
Carbocyclic nucleosides are of considerable interest for the development of new therapeutic agents. A key reaction in the preparation of many such nucleoside analogues is dihydroxylation of appropriately substituted cyclopentenes. Although often considered a routine reaction, in this paper, we report the dramatic influence of substituents on the facial selectivity of dihydroxylations. The substituted cyclopentene substrates are derived from acylnitroso cycloaddition reactions of cyclopentadiene, followed by N-O reduction and efficient enzymatic resolution. The results are directly utilized in a very efficient asymmetric synthesis of an antiviral carbocyclic nucleoside, noraristeromycin 5. Extensions toward the synthesis of carbocyclic sinefungin 7 document the importance of realizing the substituent dependence of the dihydroxylation reaction.
Figures
Similar articles
-
Highly stereoselective synthesis of aristeromycin through dihydroxylation of 4-aryl-1-azido-2-cyclopentenes.J Org Chem. 2004 Feb 6;69(3):655-9. doi: 10.1021/jo034672u. J Org Chem. 2004. PMID: 14750788
-
Synthesis and antiviral activity of carbocyclic nucleosides incorporating a modified cyclopentane ring. Part 3: Adenosine and uridine analogues.Nucleosides Nucleotides. 1999 Oct;18(10):2253-63. doi: 10.1080/07328319908044879. Nucleosides Nucleotides. 1999. PMID: 10616729
-
Stereoselective synthetic approaches to highly substituted cyclopentanes via electrophilic additions to mono-, di-, and trisubstituted cyclopentenes.J Org Chem. 2000 Jun 30;65(13):4058-69. doi: 10.1021/jo000101f. J Org Chem. 2000. PMID: 10866624
-
Chemical Approaches to Carbocyclic Nucleosides.Chem Rec. 2022 May;22(5):e202100307. doi: 10.1002/tcr.202100307. Epub 2022 Feb 4. Chem Rec. 2022. PMID: 35119191 Review.
-
Recent progress for the synthesis of selected carbocyclic nucleosides.Future Med Chem. 2015;7(13):1809-28. doi: 10.4155/fmc.15.105. Epub 2015 Sep 29. Future Med Chem. 2015. PMID: 26416300 Review.
Cited by
-
Synthesis of carbapyochelins via diastereoselective azidation of 5-(ethoxycarbonyl)methylproline derivatives.J Org Chem. 2008 Sep 19;73(18):7420-3. doi: 10.1021/jo801294p. Epub 2008 Aug 13. J Org Chem. 2008. PMID: 18698823 Free PMC article.
-
Syntheses of carbocyclic aminonucleosides and (-)-epi-4'-carbocyclic puromycin: Application of palladium(0)/indium iodide-allylations and tethered aminohydroxylations.Tetrahedron Lett. 2010 Jun 9;51(23):3053-3056. doi: 10.1016/j.tetlet.2010.04.006. Tetrahedron Lett. 2010. PMID: 20640052 Free PMC article.
-
Syntheses of new spirocarbocyclic nucleoside analogs using iminonitroso Diels-Alder reactions.Org Lett. 2009 Jan 15;11(2):449-52. doi: 10.1021/ol802553g. Org Lett. 2009. PMID: 19072701 Free PMC article.
-
Titanocene(III) chloride-mediated reductions of oxazines, hydroxamic acids, and N-hydroxy carbamates.J Org Chem. 2009 Jan 2;74(1):448-51. doi: 10.1021/jo802184y. J Org Chem. 2009. PMID: 19053586 Free PMC article.
-
Diastereoselective synthesis of a spironoraristeromycin using an acylnitroso Diels-Alder reaction.J Org Chem. 2009 Aug 21;74(16):5941-6. doi: 10.1021/jo900877b. J Org Chem. 2009. PMID: 19601571 Free PMC article.
References
-
-
For a discussion of inhibitors of enzymes involved with purine and pyrimidine metabolism, see: Hobbs JB. In: Comprehensive Medicinal Chemistry. Hansch C, editor. vol. 2. Pergamon; New York: 1990. p. 306..
-
-
- Trost BM, Madsen R, Guile SD, Brown B. J. Am. Chem. Soc. 2000;122:5947.
- Palmer CF, McCague R, Ruecroft G, Savage S, Taylor SJC, Ries C. Tetrahedron Lett. 1996;37:4601.
- Li F, Brogan JB, Gage JL, Zhang D, Miller MJ. J. Org. Chem. 2004;69:4538. - PubMed
-
- Goodchild J. In: Topics in Antibiotic Chemistry. Sammes PG, editor. vol. 6B. Wiley; New York: 1982. p. 99.
-
- Bennett LL, Jr., Brockman RW, Rose LM, Allan PW, Shaddix SC, Shealy YF, Clayton JD. Mol. Pharmacol. 1985;27:666. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources