Rhamnolipids are virulence factors that promote early infiltration of primary human airway epithelia by Pseudomonas aeruginosa
- PMID: 16714541
- PMCID: PMC1479292
- DOI: 10.1128/IAI.01772-05
Rhamnolipids are virulence factors that promote early infiltration of primary human airway epithelia by Pseudomonas aeruginosa
Abstract
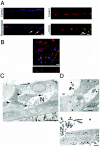
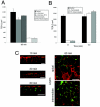
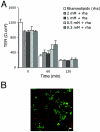
The opportunistic bacterium Pseudomonas aeruginosa causes chronic respiratory infections in cystic fibrosis and immunocompromised individuals. Bacterial adherence to the basolateral domain of the host cells and internalization are thought to participate in P. aeruginosa pathogenicity. However, the mechanism by which the pathogen initially modulates the paracellular permeability of polarized respiratory epithelia remains to be understood. To investigate this mechanism, we have searched for virulence factors secreted by P. aeruginosa that affect the structure of human airway epithelium in the early stages of infection. We have found that only bacterial strains secreting rhamnolipids were efficient in modulating the barrier function of an in vitro-reconstituted human respiratory epithelium, irrespective of their release of elastase and lipopolysaccharide. In contrast to previous reports, we document that P. aeruginosa was not internalized by epithelial cells. We further report that purified rhamnolipids, applied on the surfaces of the epithelia, were sufficient to functionally disrupt the epithelia and to promote the paracellular invasion of rhamnolipid-deficient P. aeruginosa. The mechanism involves the incorporation of rhamnolipids within the host cell membrane, leading to tight-junction alterations. The study provides direct evidence for a hitherto unknown mechanism whereby the junction-dependent barrier of the respiratory epithelium is selectively altered by rhamnolipids.
Figures





Comment in
-
Airway epithelial (nasal) cell monolayers used to study Pseudomonas aeruginosa invasion are hyperpolarized and not representative of the human airway epithelium.Infect Immun. 2006 Dec;74(12):7043; author reply 7043-4. doi: 10.1128/IAI.01233-06. Infect Immun. 2006. PMID: 17116966 Free PMC article. No abstract available.
References
-
- Avdeef, A. 2001. Physicochemical profiling (solubility, permeability and charge state). Curr. Top. Med. Chem. 1:277-351. - PubMed
-
- Azghani, A. O. 1996. Pseudomonas aeruginosa and epithelial permeability: a role of virulence factors elastase and exotoxin A. Am. J. Respir. Cell Mol. Biol. 15:132-140. - PubMed
-
- Azghani, A. O., T. Bedinghaus, and R. Klein. 2000. Detection of elastase from Pseudomonas aeruginosa in sputum and its potential role in epithelial cell permeability. Lung 178:181-189. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

