Genetic variation among isolates of Sarcocystis neurona, the agent of protozoal myeloencephalitis, as revealed by amplified fragment length polymorphism markers
- PMID: 16714575
- PMCID: PMC1479295
- DOI: 10.1128/IAI.01215-05
Genetic variation among isolates of Sarcocystis neurona, the agent of protozoal myeloencephalitis, as revealed by amplified fragment length polymorphism markers
Abstract
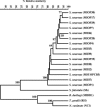
Sarcocystis neurona causes serious neurological disease in horses and other vertebrates in the Americas. Based on epidemiological data, this parasite has recently emerged. Here, the genetic diversity of Sarcocystis neurona was evaluated using the amplified fragment length polymorphism (AFLP) method. Fifteen S. neurona taxa from different regions collected over the last 10 years were used; six isolates were from clinically diseased horses, eight isolates were from wild-caught opossums (Didelphis virginiana), and one isolate was from a cowbird (Molothrus ater). Additionally, four outgroup taxa were also fingerprinted. Nine primer pairs were used to generate AFLP patterns, with a total number of amplified fragments ranging from 30 to 60, depending on the isolate and primers tested. Based on the presence/absence of amplified AFLP fragments and pairwise similarity values, all the S. neurona isolates tested were clustered in one monophyletic group. No significant correlation could be found between genomic similarity and host origin of the S. neurona isolates. AFLP revealed significant intraspecific genetic variations, and S. neurona appeared as a highly variable species. Furthermore, linkage disequilibrium analysis suggested that S. neurona populations within Michigan have an intermediate type of population structure that includes characteristics of both clonal and panamictic population structures. AFLP is a reliable molecular technique that has provided one of the most informative approaches to ascertain phylogenetic relationships in S. neurona and its closest relatives, allowing them to be clustered by relative similarity using band matching and unweighted pair group method with arithmetic mean analysis, which may be applicable to other related protozoal species.
Figures
Similar articles
-
Amplified fragment length polymorphism: an adept technique for genome mapping, genetic differentiation, and intraspecific variation in protozoan parasites.Parasitol Res. 2013 Feb;112(2):457-66. doi: 10.1007/s00436-012-3238-6. Epub 2012 Dec 20. Parasitol Res. 2013. PMID: 23254590 Review.
-
Brown-headed cowbirds (Molothrus ater) harbor Sarcocystis neurona and act as intermediate hosts.Vet Parasitol. 2008 May 6;153(1-2):24-43. doi: 10.1016/j.vetpar.2007.12.016. Epub 2007 Dec 23. Vet Parasitol. 2008. PMID: 18342449
-
Evidence to support horses as natural intermediate hosts for Sarcocystis neurona.Vet Parasitol. 2005 Oct 10;133(1):27-36. doi: 10.1016/j.vetpar.2005.05.016. Vet Parasitol. 2005. PMID: 15970386
-
Sarcocystis falcatula from passerine and psittacine birds: synonymy with Sarcocystis neurona, agent of equine protozoal myeloencephalitis.J Parasitol. 1995 Dec;81(6):930-5. J Parasitol. 1995. PMID: 8544067
-
Molecular typing of Sarcocystis neurona: current status and future trends.Vet Parasitol. 2007 Oct 21;149(1-2):43-55. doi: 10.1016/j.vetpar.2007.06.039. Epub 2007 Aug 16. Vet Parasitol. 2007. PMID: 17706872 Review.
Cited by
-
Limited genetic diversity among Sarcocystis neurona strains infecting southern sea otters precludes distinction between marine and terrestrial isolates.Vet Parasitol. 2010 Apr 19;169(1-2):37-44. doi: 10.1016/j.vetpar.2009.12.020. Epub 2009 Dec 22. Vet Parasitol. 2010. PMID: 20071081 Free PMC article.
-
An update on Sarcocystis neurona infections in animals and equine protozoal myeloencephalitis (EPM).Vet Parasitol. 2015 Apr 15;209(1-2):1-42. doi: 10.1016/j.vetpar.2015.01.026. Epub 2015 Feb 7. Vet Parasitol. 2015. PMID: 25737052 Free PMC article. Review.
-
Amplified fragment length polymorphism: an adept technique for genome mapping, genetic differentiation, and intraspecific variation in protozoan parasites.Parasitol Res. 2013 Feb;112(2):457-66. doi: 10.1007/s00436-012-3238-6. Epub 2012 Dec 20. Parasitol Res. 2013. PMID: 23254590 Review.
-
Self-mating in the definitive host potentiates clonal outbreaks of the apicomplexan parasites Sarcocystis neurona and Toxoplasma gondii.PLoS Genet. 2010 Dec 23;6(12):e1001261. doi: 10.1371/journal.pgen.1001261. PLoS Genet. 2010. PMID: 21203443 Free PMC article.
-
Equine Protozoal Myeloencephalitis: An Updated Consensus Statement with a Focus on Parasite Biology, Diagnosis, Treatment, and Prevention.J Vet Intern Med. 2016 Mar-Apr;30(2):491-502. doi: 10.1111/jvim.13834. Epub 2016 Feb 9. J Vet Intern Med. 2016. PMID: 26857902 Free PMC article. Review.
References
-
- Blears, M. J., N. J. Pokorny, R. A. Carreno, S. Chen, S. A. De Grandis, H. Lee, and J. T. Trevors. 2000. DNA fingerprinting of Cryptosporidium parvum isolates using amplified fragment length polymorphism (AFLP). J. Parasitol. 86:838-841. - PubMed
-
- Cheadle, M. A., S. M. Tanhauser, J. B. Dame, D. C. Sellon, M. Hines, P. E. Ginn, R. J. MacKay, and E. C. Greiner. 2001. The nine-banded armadillo (Dasypus novemcinctus) is an intermediate host for Sarcocystis neurona. Int. J. Parasitol. 31:330-335. - PubMed
-
- Cheadle, M. A., C. A. Yowell, D. C. Sellon, M. Hines, P. E. Ginn, A. E. Marsh, R. J. MacKay, J. B. Dame, and E. C. Greiner. 2001. The striped skunk (Mephitis mephitis) is an intermediate host for Sarcocystis neurona. Int. J. Parasitol. 31:843-849. - PubMed
-
- Dubey, J. P., S. S. Black, L. G. Rickard, B. M. Rosenthal, D. S. Lindsay, S. K. Shen, O. C. Kwok, G. Hurst, and A. Rashmir-Raven. 2001. Prevalence of Sarcocystis neurona sporocysts in opossums (Didelphis virginiana) from rural Mississippi. Vet. Parasitol. 95:283-293. - PubMed
-
- Dubey, J. P., D. S. Lindsay, W. J. Saville, S. M. Reed, D. E. Granstrom, and C. A. Speer. 2001. A review of Sarcocystis neurona and equine protozoal myeloencephalitis (EPM). Vet. Parasitol. 95:89-131. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

