Cost-effectiveness analysis of colorectal cancer screening with stool DNA testing in intermediate-incidence countries
- PMID: 16723013
- PMCID: PMC1525200
- DOI: 10.1186/1471-2407-6-136
Cost-effectiveness analysis of colorectal cancer screening with stool DNA testing in intermediate-incidence countries
Abstract
Background: The aim of this study is to compare the cost-effectiveness of screening with stool DNA testing with that of screening with other tools (annual fecal occult blood testing, flexible sigmoidoscopy every 5 years, and colonoscopy every 10 years) or not screening at all.
Methods: We developed a Markov model to evaluate the above screening strategies in the general population 50 to 75 years of age in Taiwan. Sensitivity analyses were performed to assess the influence of various parameters on the cost-effectiveness of screening. A third-party payer perspective was adopted and the cost of dollar 13,000 per life-year saved (which is roughly the per capita GNP of Taiwan in 2003) was chosen as the ceiling ratio for assessing whether the program is cost-effective.
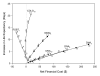
Results: Stool DNA testing every three, five, and ten years can reduce colorectal cancer mortality by 22%, 15%, and 9%, respectively. The associated incremental costs were dollar 9,794, dollar 9,335, and dollar 7,717, per life-year saved when compared with no screening. Stool DNA testing strategies were the least cost-effective with the cost per stool DNA test, referral rate with diagnostic colonoscopy, prevalence of large adenoma, and discount rate being the most influential parameters.
Conclusion: In countries with a low or intermediate incidence of colorectal cancer, stool DNA testing is less cost-effective than the other currently recommended strategies for population-based screening, particularly targeting at asymptomatic subjects.
Figures
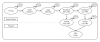
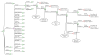
 : Markov cycle. Abbreviations: CRC, colorectal cancer; DNA3, stool DNA testing every 3 years; DNA5, stool DNA testing every 5 years; DNA10, stool DNA testing every 10 years; FOBT1, fecal occult blood testing every year; SIGM5, sigmoidoscopy every 5 years; COLO10, colonoscopy every 10 years; Small adenoma, adenoma smaller than 1 cm in size; large adenoma, adenoma larger than 1 cm in size; early CRC, Dukes' stage A and B colorectal cancer; late CRC, Dukes' stage C and D colorectal cancer.
: Markov cycle. Abbreviations: CRC, colorectal cancer; DNA3, stool DNA testing every 3 years; DNA5, stool DNA testing every 5 years; DNA10, stool DNA testing every 10 years; FOBT1, fecal occult blood testing every year; SIGM5, sigmoidoscopy every 5 years; COLO10, colonoscopy every 10 years; Small adenoma, adenoma smaller than 1 cm in size; large adenoma, adenoma larger than 1 cm in size; early CRC, Dukes' stage A and B colorectal cancer; late CRC, Dukes' stage C and D colorectal cancer.
Similar articles
-
Comparative effectiveness and cost-effectiveness of screening colonoscopy vs. sigmoidoscopy and alternative strategies.Am J Gastroenterol. 2013 Jan;108(1):120-32. doi: 10.1038/ajg.2012.380. Epub 2012 Dec 18. Am J Gastroenterol. 2013. PMID: 23247579
-
Cost-effectiveness analysis on screening for colorectal neoplasm and management of colorectal cancer in Asia.Aliment Pharmacol Ther. 2008 Aug 1;28(3):353-63. doi: 10.1111/j.1365-2036.2008.03726.x. Aliment Pharmacol Ther. 2008. PMID: 18638075
-
Cost effectiveness and projected national impact of colorectal cancer screening in France.Endoscopy. 2011 Sep;43(9):780-93. doi: 10.1055/s-0030-1256409. Epub 2011 May 27. Endoscopy. 2011. PMID: 21623557
-
[Individual screening for colorectal cancer: which strategy for which patient?].Rev Med Brux. 2001 Sep;22(4):A203-9. Rev Med Brux. 2001. PMID: 11680174 Review. French.
-
Cutting cost and increasing access to colorectal cancer screening: another approach to following the guidelines.Cancer Epidemiol Biomarkers Prev. 2006 Jan;15(1):108-13. doi: 10.1158/1055-9965.EPI-05-0198. Cancer Epidemiol Biomarkers Prev. 2006. PMID: 16434595 Review.
Cited by
-
Fecal DNA screening in colorectal cancer.Can J Gastroenterol. 2008 Jul;22(7):631-3. doi: 10.1155/2008/761208. Can J Gastroenterol. 2008. PMID: 18629393 Free PMC article. Review.
-
Cost-effectiveness of colorectal cancer screening - an overview.Best Pract Res Clin Gastroenterol. 2010 Aug;24(4):439-49. doi: 10.1016/j.bpg.2010.04.004. Best Pract Res Clin Gastroenterol. 2010. PMID: 20833348 Free PMC article.
-
Cost-effectiveness of colorectal cancer screening.Epidemiol Rev. 2011;33(1):88-100. doi: 10.1093/epirev/mxr004. Epub 2011 Jun 1. Epidemiol Rev. 2011. PMID: 21633092 Free PMC article. Review.
-
Cost-effectiveness of colorectal cancer screening protocols in urban Chinese populations.PLoS One. 2014 Oct 6;9(10):e109150. doi: 10.1371/journal.pone.0109150. eCollection 2014. PLoS One. 2014. PMID: 25285526 Free PMC article.
-
DNA methylation analysis of SFRP2, GATA4/5, NDRG4 and VIM for the detection of colorectal cancer in fecal DNA.Oncol Lett. 2014 Oct;8(4):1751-1756. doi: 10.3892/ol.2014.2413. Epub 2014 Aug 4. Oncol Lett. 2014. PMID: 25202404 Free PMC article.
References
-
- Rudy DR, Zdon MJ. Update on colorectal cancer. Am Fam Physician. 2000;61:1759–1770. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

