Dissociation of a MAVS/IPS-1/VISA/Cardif-IKKepsilon molecular complex from the mitochondrial outer membrane by hepatitis C virus NS3-4A proteolytic cleavage
- PMID: 16731946
- PMCID: PMC1472616
- DOI: 10.1128/JVI.02495-05
Dissociation of a MAVS/IPS-1/VISA/Cardif-IKKepsilon molecular complex from the mitochondrial outer membrane by hepatitis C virus NS3-4A proteolytic cleavage
Abstract
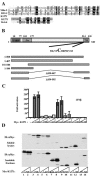
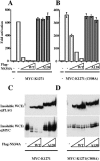
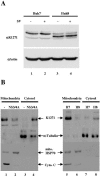
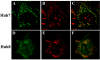
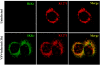
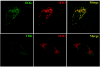
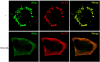
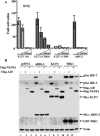
Intracellular RNA virus infection is detected by the cytoplasmic RNA helicase RIG-I that plays an essential role in signaling to the host antiviral response. Recently, the adapter molecule that links RIG-I sensing of incoming viral RNA to downstream signaling and gene activation events was characterized by four different groups; MAVS/IPS-1-1/VISA/Cardif contains an amino-terminal CARD domain and a carboxyl-terminal mitochondrial transmembrane sequence that localizes to the mitochondrial membrane. Furthermore, the hepatitis C virus NS3-4A protease complex specifically targets MAVS/IPS-1/VISA/Cardif for cleavage as part of its immune evasion strategy. With a novel search program written in python, we also identified an uncharacterized protein, KIAA1271 (K1271), containing a single CARD-like domain at the N terminus and a Leu-Val-rich C terminus that is identical to that of MAVS/IPS-1/VISA/Cardif. Using a combination of biochemical analysis, subcellular fractionation, and confocal microscopy, we now demonstrate that NS3-4A cleavage of MAVS/IPS-1/VISA/Cardif/K1271 results in its dissociation from the mitochondrial membrane and disrupts signaling to the antiviral immune response. Furthermore, virus-induced IKKepsilon kinase, but not TBK1, colocalized strongly with MAVS at the mitochondrial membrane, and the localization of both molecules was disrupted by NS3-4A expression. Mutation of the critical cysteine 508 to alanine was sufficient to maintain mitochondrial localization of MAVS/IPS-1/VISA/Cardif and IKKepsilon in the presence of NS3-4A. These observations provide an outline of the mechanism by which hepatitis C virus evades the interferon antiviral response.
Figures

Similar articles
-
Recruitment of an interferon molecular signaling complex to the mitochondrial membrane: disruption by hepatitis C virus NS3-4A protease.Biochem Pharmacol. 2006 Nov 30;72(11):1477-84. doi: 10.1016/j.bcp.2006.06.030. Epub 2006 Jul 31. Biochem Pharmacol. 2006. PMID: 16876765
-
Hepatitis C virus NS2 and NS3/4A proteins are potent inhibitors of host cell cytokine/chemokine gene expression.Virol J. 2006 Sep 1;3:66. doi: 10.1186/1743-422X-3-66. Virol J. 2006. PMID: 16945160 Free PMC article.
-
MasterCARD: a priceless link to innate immunity.Trends Mol Med. 2006 Feb;12(2):53-6. doi: 10.1016/j.molmed.2005.12.003. Epub 2006 Jan 6. Trends Mol Med. 2006. PMID: 16406812 Review.
-
Induction of IRF-3 and IRF-7 phosphorylation following activation of the RIG-I pathway.Cell Mol Biol (Noisy-le-grand). 2006 May 15;52(1):17-28. Cell Mol Biol (Noisy-le-grand). 2006. PMID: 16914100
-
The interferon inducing pathways and the hepatitis C virus.World J Gastroenterol. 2007 May 7;13(17):2446-54. doi: 10.3748/wjg.v13.i17.2446. World J Gastroenterol. 2007. PMID: 17552028 Free PMC article. Review.
Cited by
-
Proteomic analysis of mitochondrial-associated ER membranes (MAM) during RNA virus infection reveals dynamic changes in protein and organelle trafficking.PLoS One. 2015 Mar 3;10(3):e0117963. doi: 10.1371/journal.pone.0117963. eCollection 2015. PLoS One. 2015. PMID: 25734423 Free PMC article.
-
Viral evasion and subversion of pattern-recognition receptor signalling.Nat Rev Immunol. 2008 Dec;8(12):911-22. doi: 10.1038/nri2436. Nat Rev Immunol. 2008. PMID: 18989317 Free PMC article. Review.
-
The structural protein VP3 of enterovirus D68 interacts with MAVS to inhibit the NF-κB signaling pathway.J Virol. 2025 Apr 15;99(4):e0016325. doi: 10.1128/jvi.00163-25. Epub 2025 Mar 5. J Virol. 2025. PMID: 40042308 Free PMC article.
-
Hepatitis C virus NS2 coordinates virus particle assembly through physical interactions with the E1-E2 glycoprotein and NS3-NS4A enzyme complexes.J Virol. 2011 Feb;85(4):1706-17. doi: 10.1128/JVI.02268-10. Epub 2010 Dec 8. J Virol. 2011. PMID: 21147927 Free PMC article.
-
A Multiantigenic DNA Vaccine That Induces Broad Hepatitis C Virus-Specific T-Cell Responses in Mice.J Virol. 2015 Aug;89(15):7991-8002. doi: 10.1128/JVI.00803-15. Epub 2015 May 27. J Virol. 2015. PMID: 26018154 Free PMC article.
References
-
- Akira, S., and K. Takeda. 2004. Toll-like receptor signalling. Nat. Rev. Immunol. 4:499-511. - PubMed
-
- Alexopoulou, L., A. C. Holt, R. Medzhitov, and R. A. Flavell. 2001. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 413:732-738. - PubMed
-
- Alter, H. J., and L. B. Seeff. 2000. Recovery, persistence, and sequelae in hepatitis C virus infection: a perspective on long-term outcome. Semin. Liver Dis. 20:17-35. - PubMed
-
- Blight, K. J., A. A. Kolykhalov, and C. M. Rice. 2000. Efficient initiation of HCV RNA replication in cell culture. Science 290:1972-1974. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

