Splice-site A choice targets plasma-membrane Ca2+-ATPase isoform 2 to hair bundles
- PMID: 16763025
- PMCID: PMC2204085
- DOI: 10.1523/JNEUROSCI.0447-06.2006
Splice-site A choice targets plasma-membrane Ca2+-ATPase isoform 2 to hair bundles
Abstract
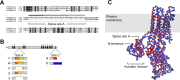
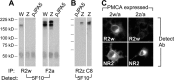
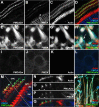
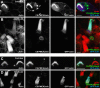
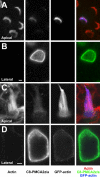
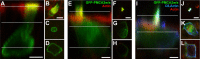
Localization of mechanotransduction in sensory hair cells to hair bundles requires selective targeting of essential proteins to specific locations. Isoform 2 of the plasma-membrane Ca2+-ATPase (PMCA2), required for hearing and balance, is found exclusively in hair bundles. We determined the contribution of splicing at the two major splicing sites (A and C) to hair-cell targeting of PMCA2. When PMCA2 isoforms were immunoprecipitated from purified hair bundles of rat utricle, 2w was the only site A variant detected; moreover, immunocytochemistry for 2w in rat vestibular and cochlear tissues indicated that this splice form was located solely in bundles. To demonstrate the necessity of the 2w sequence, we transfected hair cells with PMCA2 containing different variants at splice sites A and C. Although native hair bundles exclusively use the 2a form at splice-site C, epitope-tagged PMCA2w/a and PMCA2w/b were both concentrated in bundles, indicating that site C is not involved in bundle targeting. In contrast, PMCA2z/a was excluded from bundles and was instead targeted to the basolateral plasma membrane. Bundle-specific targeting of PMCA2w/a tagged with green fluorescent protein (GFP) was diminished, suggesting that GFP interfered with splice-site A. Together, these data demonstrate that PMCA2w/a is the hair-bundle isoform of PMCA in rat hair cells and that 2w targets PMCA2 to bundles. The 2w sequence is thus the first targeting signal identified for a hair-bundle membrane protein; moreover, the striking distribution of inner-ear PMCA isoforms dictated by selective targeting suggests a critical functional role for segregated pathways of Ca2+ transport.
Figures

Similar articles
-
Plasma membrane Ca2+-ATPase isoform 2a is the PMCA of hair bundles.J Neurosci. 2001 Jul 15;21(14):5066-78. doi: 10.1523/JNEUROSCI.21-14-05066.2001. J Neurosci. 2001. PMID: 11438582 Free PMC article.
-
Alternative splicing of the first intracellular loop of plasma membrane Ca2+-ATPase isoform 2 alters its membrane targeting.J Biol Chem. 2003 May 16;278(20):18464-70. doi: 10.1074/jbc.M301482200. Epub 2003 Mar 6. J Biol Chem. 2003. PMID: 12624087
-
Rapid turnover of stereocilia membrane proteins: evidence from the trafficking and mobility of plasma membrane Ca(2+)-ATPase 2.J Neurosci. 2006 Jun 7;26(23):6386-95. doi: 10.1523/JNEUROSCI.1215-06.2006. J Neurosci. 2006. PMID: 16763047 Free PMC article.
-
The plasma membrane calcium pump in the hearing process: physiology and pathology.Sci China Life Sci. 2011 Aug;54(8):686-90. doi: 10.1007/s11427-011-4200-z. Epub 2011 Jul 24. Sci China Life Sci. 2011. PMID: 21786191 Review.
-
Role of alternative splicing in generating isoform diversity among plasma membrane calcium pumps.Physiol Rev. 2001 Jan;81(1):21-50. doi: 10.1152/physrev.2001.81.1.21. Physiol Rev. 2001. PMID: 11152753 Review.
Cited by
-
The novel mouse mutation Oblivion inactivates the PMCA2 pump and causes progressive hearing loss.PLoS Genet. 2008 Oct;4(10):e1000238. doi: 10.1371/journal.pgen.1000238. Epub 2008 Oct 31. PLoS Genet. 2008. PMID: 18974863 Free PMC article.
-
Apical localization of PMCA2w/b is enhanced in terminally polarized MDCK cells.Biochem Biophys Res Commun. 2011 Jul 1;410(2):322-7. doi: 10.1016/j.bbrc.2011.05.147. Epub 2011 Jun 6. Biochem Biophys Res Commun. 2011. PMID: 21672522 Free PMC article.
-
Plasma membrane calcium pump (PMCA) isoform 4 is targeted to the apical membrane by the w-splice insert from PMCA2.Cell Calcium. 2012 Feb;51(2):171-8. doi: 10.1016/j.ceca.2011.12.010. Epub 2012 Jan 16. Cell Calcium. 2012. PMID: 22252018 Free PMC article.
-
PDZD7-MYO7A complex identified in enriched stereocilia membranes.Elife. 2016 Aug 15;5:e18312. doi: 10.7554/eLife.18312. Elife. 2016. PMID: 27525485 Free PMC article.
-
Differential expression of PMCA2 mRNA isoforms in a cohort of Spanish patients with breast tumor types.Oncol Lett. 2018 Dec;16(6):6950-6959. doi: 10.3892/ol.2018.9540. Epub 2018 Oct 2. Oncol Lett. 2018. PMID: 30546427 Free PMC article.
References
-
- Abacioglu YH, Fouts TR, Laman JD, Claassen E, Pincus SH, Moore JP, Roby CA, Kamin-Lewis R, Lewis GK (1994). Epitope mapping and topology of baculovirus-expressed HIV-1 gp160 determined with a panel of murine monoclonal antibodies. AIDS Res Hum Retroviruses 10:371–381. - PubMed
-
- Anniko M, Lim D, Wroblewski R (1984). Elemental composition of individual cells and tissues in the cochlea. Acta Otolaryngol 98:439–453. - PubMed
-
- Belyantseva IA, Boger ET, Naz S, Frolenkov GI, Sellers JR, Ahmed ZM, Griffith AJ, Friedman TB (2005). Myosin-XVa is required for tip localization of whirlin and differential elongation of hair-cell stereocilia. Nat Cell Biol 7:148–156. - PubMed
-
- Blaustein MP, Lederer WJ (1999). Sodium/calcium exchange: its physiological implications. Physiol Rev 79:763–854. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
