Concomitant deficits in working memory and fear extinction are functionally dissociated from reduced anxiety in metabotropic glutamate receptor 7-deficient mice
- PMID: 16775145
- PMCID: PMC6674050
- DOI: 10.1523/JNEUROSCI.1497-06.2006
Concomitant deficits in working memory and fear extinction are functionally dissociated from reduced anxiety in metabotropic glutamate receptor 7-deficient mice
Abstract
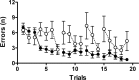
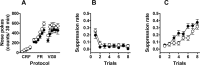
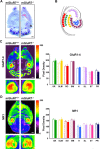
Metabotropic glutamate receptor 7 (mGluR7), a receptor with a distinct brain distribution and a putative role in anxiety, emotional responding, and spatial working memory, could be an interesting therapeutic target for fear and anxiety disorders. mGluR7-deficient (mGluR7-/-) mice showed essentially normal performance in tests for neuromotor and exploratory activity and passive avoidance learning but prominent anxiolytic behavior in two anxiety tests. They showed a delayed learning curve during the acquisition of the hidden-platform water maze, and three interspersed probe trials indicated that mGluR7-/- mice were slower to acquire spatial information. Working memory in the water maze task and the radial arm maze was impaired in mGluR7-/- mice compared with mGluR7+/+. mGluR7-/- mice also displayed a higher resistance to extinction of fear-elicited response suppression in a conditioned emotional response protocol. In a non-fear-based water maze protocol, mGluR7-/- mice displayed similar delayed extinction. These observed behavioral changes are probably not attributable to changes in AMPA or NMDA receptor function because expression levels of AMPA and NMDA receptors were unaltered. Extinction of conditioned fear is an active and context-dependent form of inhibitory learning and an experimental model for therapeutic fear reduction. It appears to depend on glutamatergic and higher-level brain functions similar to those involved in spatial working memory but functionally dissociated from those that mediate constitutional responses in anxiety tests.
Figures
Similar articles
-
Deficits in acquisition and extinction of conditioned responses in mGluR7 knockout mice.Neurobiol Learn Mem. 2008 Jul;90(1):103-11. doi: 10.1016/j.nlm.2008.01.001. Epub 2008 Mar 4. Neurobiol Learn Mem. 2008. PMID: 18289889
-
Antidepressant and anxiolytic-like effects in mice lacking the group III metabotropic glutamate receptor mGluR7.Eur J Neurosci. 2003 Jun;17(11):2409-17. doi: 10.1046/j.1460-9568.2003.02667.x. Eur J Neurosci. 2003. PMID: 12814372
-
NMDA receptor subunit NR2A is required for rapidly acquired spatial working memory but not incremental spatial reference memory.J Neurosci. 2008 Apr 2;28(14):3623-30. doi: 10.1523/JNEUROSCI.3639-07.2008. J Neurosci. 2008. PMID: 18385321 Free PMC article.
-
Glutamate receptors in extinction and extinction-based therapies for psychiatric illness.Neuropsychopharmacology. 2011 Jan;36(1):274-93. doi: 10.1038/npp.2010.88. Epub 2010 Jul 14. Neuropsychopharmacology. 2011. PMID: 20631689 Free PMC article. Review.
-
Metabotropic glutamate receptor subtype 5 antagonism in learning and memory.Eur J Pharmacol. 2010 Aug 10;639(1-3):17-25. doi: 10.1016/j.ejphar.2009.12.039. Epub 2010 Apr 2. Eur J Pharmacol. 2010. PMID: 20363219 Free PMC article. Review.
Cited by
-
A20/TNFAIP3 heterozygosity predisposes to behavioral symptoms in a mouse model for neuropsychiatric lupus.Brain Behav Immun Health. 2019 Dec 14;2:100018. doi: 10.1016/j.bbih.2019.100018. eCollection 2020 Feb. Brain Behav Immun Health. 2019. PMID: 38377433 Free PMC article.
-
The G Protein-Coupled Glutamate Receptors as Novel Molecular Targets in Schizophrenia Treatment-A Narrative Review.J Clin Med. 2021 Apr 2;10(7):1475. doi: 10.3390/jcm10071475. J Clin Med. 2021. PMID: 33918323 Free PMC article. Review.
-
A pilot study on glutamate receptor and carrier gene variants and risk of childhood autism spectrum.Metab Brain Dis. 2023 Oct;38(7):2477-2488. doi: 10.1007/s11011-023-01272-w. Epub 2023 Aug 14. Metab Brain Dis. 2023. PMID: 37578654
-
Antipsychotic pharmacogenomics in first episode psychosis: a role for glutamate genes.Transl Psychiatry. 2016 Feb 23;6(2):e739. doi: 10.1038/tp.2016.10. Transl Psychiatry. 2016. PMID: 26905411 Free PMC article.
-
Phenotypic profiling of mGlu7 knockout mice reveals new implications for neurodevelopmental disorders.Genes Brain Behav. 2020 Sep;19(7):e12654. doi: 10.1111/gbb.12654. Epub 2020 Apr 14. Genes Brain Behav. 2020. PMID: 32248644 Free PMC article.
References
-
- Astur RS, Taylor LB, Mamelak AN, Philpott L, Sutherland RJ (2002). Humans with hippocampus damage display severe spatial memory impairments in a virtual Morris water task. Behav Brain Res 132:77–84. - PubMed
-
- Berman DE, Dudai Y (2001). Memory extinction, learning anew, and learning the new: dissociations in the molecular machinery of learning in cortex. Science 291:2417–2419. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
